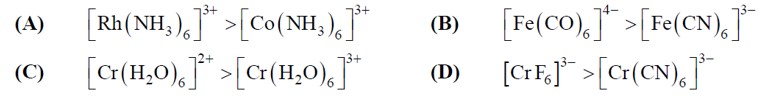9.27 What is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
9.27 What is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
The strong ligands have higher splitting power of d orbitals of the central metal ion, whereas weak ligand has relatively lower splitting power of d orbitals of the central metal ion. The energy difference between t2g and eg sets of d orbitals is CFSE. The strength of the ligands depend on the magni
Similar Questions for you
CoCl3.NH3 + AgNO3
x = 5
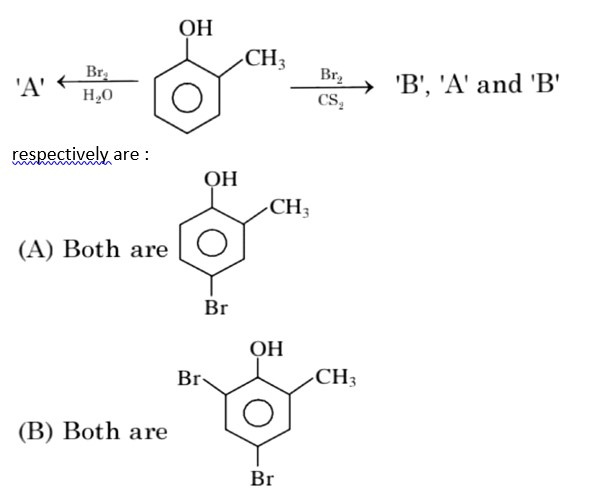
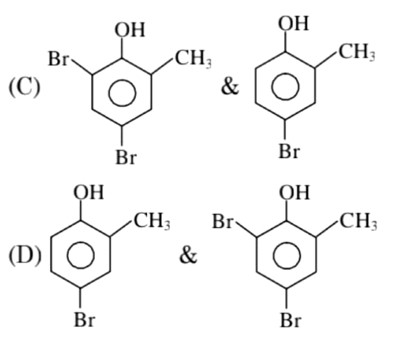
In H2O (polar solvent) dibromophenol derivative and in CS2 (non-polar solvent moneobromo phenol derivate is obtained.
3d => 4d => 5d CFSE increases for the same ligands.
Factual
⇒ leaching methods is used for those metal in which metal is more soluble than impurities and these are Al, Au, Ag, low grade Cu
σ bonded organometallic compound ⇒ M – C
σ-bond
and in π – bonded organo metallic compound
M – C
π bond
In ferrocene, there is π-bond
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering