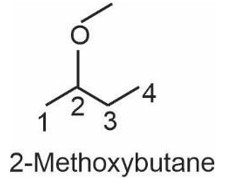
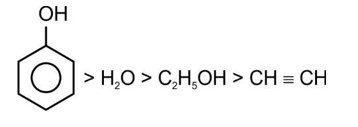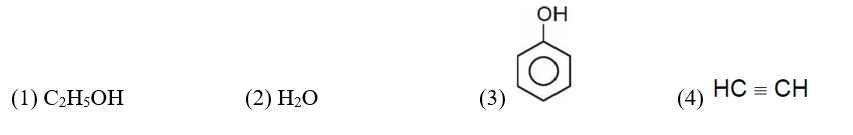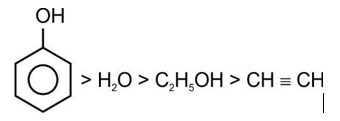11.34 Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
11.34 Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
41 Views|Posted a year ago
Asked by Shiksha User
1 Answer
A
Answered by
a year ago
11.34
Due to the presence of -OH group, ethanol undergoes intermolecular hydrogen bonding which results in the association of molecules.
Therefore, extra energy is required to break those hydrogen bonds. Whereas methoxymethane does not undergo those hydrogen bonding which implies ethanol has a higher
Similar Questions for you
Rainbow is formed due to internal reflection and dispersion.
Correct order of acidic strength
Correct order of acidic strength
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering