13.21 Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
13.21 Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Gabriel phthalimide synthesis is used for the preparation of aliphatic primary amines. It involves nucleophilic substitution (SN2) of alkyl halides by the anion formed by the phthalimide.But aryl halides do not undergo nucleophilic substitution with the anion formed by the phthalimide.
Therefore arom
Similar Questions for you
In Amines, the nitrogen atom bonds with alkyl or aryl groups replacing hydrogen, whereas in amides, the nitrogen atom bonds directly with the carbonyl group (-CO-).
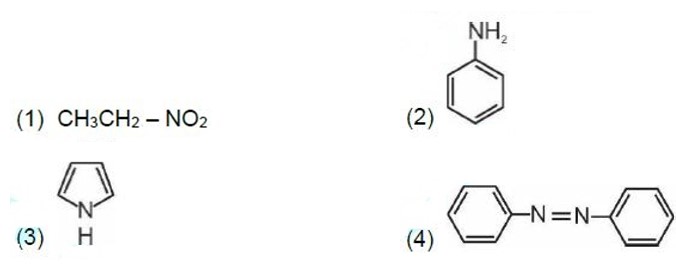
Kjeldahl's method is not applicable to compounds containing nitrogen in nitro and azo groups and nitrogen present in the ring.
Correct order of basic strength in aqueous medium is
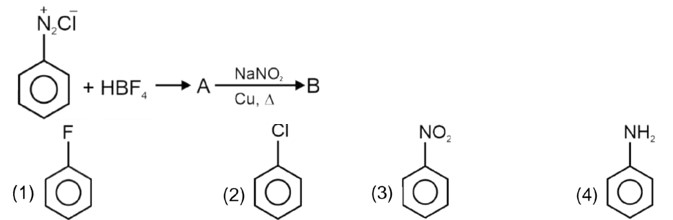
Kindly consider the following figure
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering