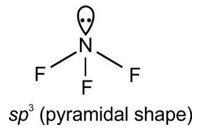
4.60. Define Lattice energy. How is Lattice energy influenced by
(i) Charge on the ions
(ii) Size of the ions?
4.60. Define Lattice energy. How is Lattice energy influenced by
(i) Charge on the ions
(ii) Size of the ions?
4.60. Lattice energy is defined as the energy released when one mole of crystalline solid is formed by the combination of oppositely charged ions.
(i) As the magnitude of charge on an ion increases there will be greater force of interionic attraction and hence greater will be the value of Lattice e
Similar Questions for you
He2 has zero bond order hence it does not exist.
The three fundamental laws of chemistry are - Law of Definite Proportions, Law of Conservation of Mass, and Law of Multiple Proportions.
The three types of chemical bonds are - ionic, metallic and covalent bonds. When the electrons transfer between the atoms, they form the Ionic bonds by producing charged ions that are attracted to each other. When atoms share electrons, covalent bonds are created. When metal atoms share a sea of del
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering