5. How is copper extracted from low grade copper ores?
5. How is copper extracted from low grade copper ores?
5. Low-grade copper ores contain a small percentage of copper that is 0.27% copper. By hydrometallurgy, low-grade copper is leached by treating with acid when copper metal goes into solution as copper ions. The solution containing copper ions is treated with scrap iron or hydrogen gas. Since iron or
Similar Questions for you
is the temperature Co-efficient of cell. The cell having less variation of EMF, with respect to temperature have high efficiency.
Below 1350° C, Mg can reduce Al2O3 and above 1350°C, Al can reduce MgO (from Ellingham diagram).
Melting and boiling point of Mg are lower than that of Al.
In ores/mineral available earthy and undesired impurities are gangue
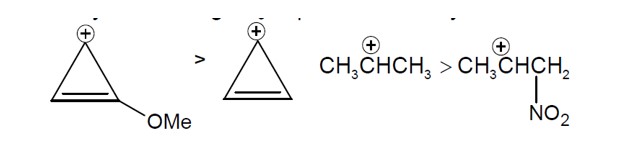
Sol. Reactivity towards depend on stability of carbocation formed
Leaching involves the given reaction,
Here, O2 is required for formation of Au (l) cyanide complex but no complex in absence of O2.
In above displacement reaction, Zn is oxidized.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 12th Chapter Six 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering