8.1. Assign oxidation number to the underlined elements in each of the following species:
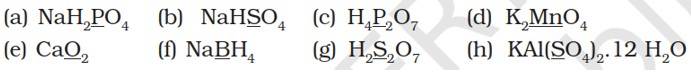
8.1. Assign oxidation number to the underlined elements in each of the following species:
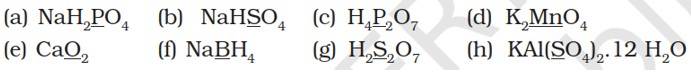
12 Views|Posted 12 months ago
Asked by Shiksha User
1 Answer
V
Answered by
12 months ago
Let x be the oxidation number to the underlined elements in the given species:
(a) NaH2PO4
(+1) + 2 (+1) + x + 4 (-2) = 0
x + 3 – 8 = 0
x = +5
(b) NaHSO4
(+1) + (+1) + x + 4 (-2) = 0
x – 6 = 0
x = +6
(c) H4P2O7
4 (+1) + 2x + 7 (-2) = 0
2x -10 =0
x = +5
(d) K2MnO4
2 (+1) + x + 4 (-2) = 0
x – 6 = 0
x = +6
(e
Similar Questions for you
Kindly go through the solution
(c) Li
Kindly go through the solution
(c) Al
Kindly go through the solution
(d) +6
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering

