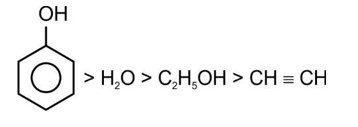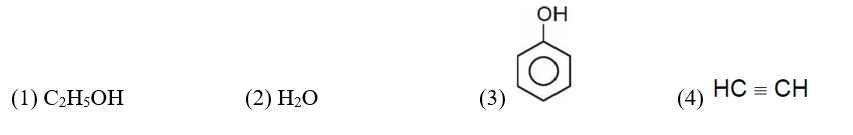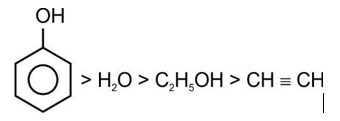A sample of 4.5mg of an unknown monohydric alcohol, R-OH was added to methylmagnesium iodide. A gas is evolved and is collected and its volume measured to be 3.1 mL. The molecular weight of the unknown alcohol is ____________ g/mol.
[Nearest integer]
A sample of 4.5mg of an unknown monohydric alcohol, R-OH was added to methylmagnesium iodide. A gas is evolved and is collected and its volume measured to be 3.1 mL. The molecular weight of the unknown alcohol is ____________ g/mol.
[Nearest integer]
3 Views|Posted 9 months ago
Asked by Shiksha User
1 Answer
P
Answered by
9 months ago
(Calculation is done considering STP condition)
No. of moles ROH = no. of moles of CH4
Similar Questions for you
Rainbow is formed due to internal reflection and dispersion.
Correct order of acidic strength
Correct order of acidic strength
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering