Among the statements , the incorrect ones are:
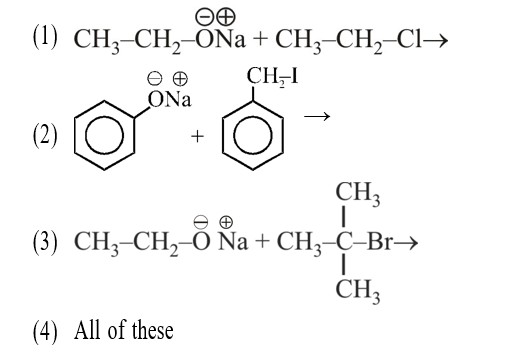
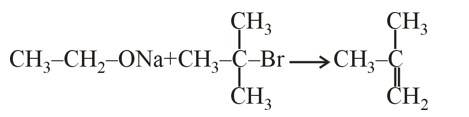
Among the statements , the incorrect ones are:
Option 1 - <p>Octahedral <span class="mathml" contenteditable="false"> <math> <mi mathvariant="normal">C</mi> <mi mathvariant="normal">o</mi> </math> </span> (III) complexes with strong field ligands have very high magnetic moments<br><!-- [if !supportLineBreakNewLine]--><br><!--[endif]--></p>
Option 2 - <p>When<span class="mathml" contenteditable="false"> <math> <msub> <mrow> <mrow> <mi mathvariant="normal">Δ</mi> </mrow> </mrow> <mrow> <mrow> <mn>0</mn> </mrow> </mrow> </msub> <mo><</mo> <mi mathvariant="normal">P</mi> </math> </span> , the d-electron configuration of <span class="mathml" contenteditable="false"> <math> <mi mathvariant="normal">C</mi> <mi mathvariant="normal">o</mi> </math> </span>(III) in an octahedral complex is <span class="mathml" contenteditable="false"> <math> <msubsup> <mrow> <mrow> <mi mathvariant="normal">t</mi> </mrow> </mrow> <mrow> <mrow> <mi mathvariant="normal">e</mi> <mi mathvariant="normal">g</mi> </mrow> </mrow> <mrow> <mrow> <mn>4</mn> </mrow> </mrow> </msubsup> <msubsup> <mrow> <mrow> <mi mathvariant="normal">e</mi> </mrow> </mrow> <mrow> <mrow> <mi mathvariant="normal">g</mi> </mrow> </mrow> <mrow> <mrow> <mn>2</mn> </mrow> </mrow> </msubsup> </math> </span><!--[endif]--><!--[endif]--></p>
Option 3 - <p>Wavelength of light absorbed by <span class="mathml" contenteditable="false"> <math> <msup> <mrow> <mrow> <mfenced open="[" close="]" separators="|"> <mrow> <mrow> <mi mathvariant="normal">C</mi> <mi mathvariant="normal">o</mi> <mo>(</mo> <mi mathvariant="normal">e</mi> <mi mathvariant="normal">n</mi> <msub> <mrow> <mrow> <mo>)</mo> </mrow> </mrow> <mrow> <mrow> <mn>3</mn> </mrow> </mrow> </msub> </mrow> </mrow> </mfenced> </mrow> </mrow> <mrow> <mrow> <mn>3</mn> <mo>+</mo> </mrow> </mrow> </msup> </math> </span> is lower than that of <span class="mathml" contenteditable="false"> <math> <msup> <mrow> <mrow> <mfenced open="[" close="]" separators="|"> <mrow> <mrow> <msub> <mrow> <mrow> <mi mathvariant="normal">C</mi> <mi mathvariant="normal">o</mi> <mi mathvariant="normal">F</mi> </mrow> </mrow> <mrow> <mrow> <mn>6</mn> </mrow> </mrow> </msub> </mrow> </mrow> </mfenced> </mrow> </mrow> <mrow> <mrow> <mn>3</mn> <mo>-</mo> </mrow> </mrow> </msup> </math> </span><!--[endif]--><!--[endif]--></p>
Option 4 - <p>If the <span class="mathml" contenteditable="false"> <math> <msub> <mrow> <mrow> <mi mathvariant="normal">Δ</mi> </mrow> </mrow> <mrow> <mrow> <mn>0</mn> </mrow> </mrow> </msub> </math> </span> for an octahedral complex of <span class="mathml" contenteditable="false"> <math> <mi mathvariant="normal">C</mi> <mi mathvariant="normal">o</mi> </math> </span> (III) is <span class="mathml" contenteditable="false"> <math> <mn>18,000</mn> <msup> <mrow> <mrow> <mtext> </mtext> <mi mathvariant="normal">c</mi> <mi mathvariant="normal">m</mi> </mrow> </mrow> <mrow> <mrow> <mo>-</mo> <mn>1</mn> </mrow> </mrow> </msup> </math> </span>, the <span class="mathml" contenteditable="false"> <math> <msub> <mrow> <mrow> <mi mathvariant="normal">Δ</mi> </mrow> </mrow> <mrow> <mrow> <mi mathvariant="normal">t</mi> </mrow> </mrow> </msub> </math> </span> for its tetrahedral complex with the same ligand will be <span class="mathml" contenteditable="false"> <math> <mn>16,000</mn> <msup> <mrow> <mrow> <mtext> </mtext> <mi mathvariant="normal">c</mi> <mi mathvariant="normal">m</mi> </mrow> </mrow> <mrow> <mrow> <mo>-</mo> <mn>1</mn> </mrow> </mrow> </msup> </math> </span><br><!-- [if !supportLineBreakNewLine]--><br><!--[endif]--></p>
2 Views|Posted 8 months ago
Asked by Shiksha User
1 Answer
A
Answered by
8 months ago
Correct Option - 4
Detailed Solution:
In strong ligand field will have of configuration and
Similar Questions for you
ΔG° = –RT * 2.303 log K
–nFE° = +RT * 2.303 log K
2 * 96500 * 0.295 = 8.314 * 298 * 2.303 log10 K
10 = log10 K = 1010
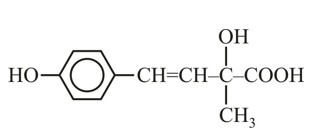
It has chiral centre and differently di substituted double bonded carbon atoms.
Rate of ESR ∝ No. of α – H (Hyperconjugation)
Cr3+ion is a most stable in aqueous solution due to. t2g half filled configuration
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Coordination Compounds 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering