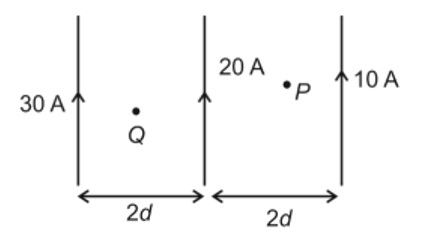
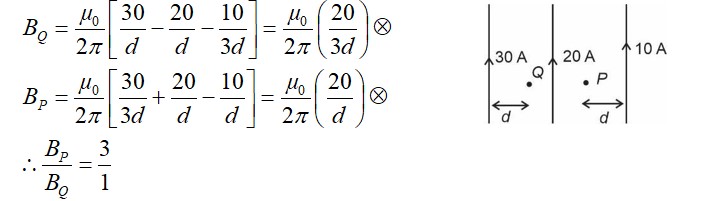Complex has a composition of . If the complex on treatment with conc. loses of its original mass, the correct molecular formula of is:
[Given : atomic mass of and ]
Complex has a composition of . If the complex on treatment with conc. loses of its original mass, the correct molecular formula of is:
[Given : atomic mass of and ]
Conc. acts as dehydrating agent.
Molar mass of given complex .
On treating with conc. the mass
lost by the complex moles of
Formula of the complex
Similar Questions for you
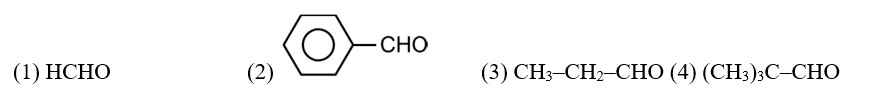
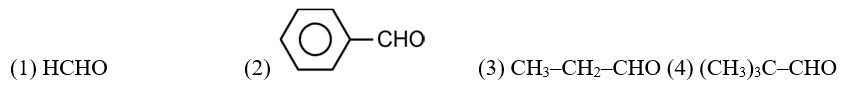
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering