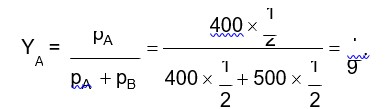Concentration terms such as mass percentage, ppm, mole fraction and molality are independent of temperature, however molarity is a function of temperature. Explain.
Concentration terms such as mass percentage, ppm, mole fraction and molality are independent of temperature, however molarity is a function of temperature. Explain.
This is a short answer type question as classified in NCERT Exemplar
The number of moles of solute dissolved in one litre of solution is the molarity of a solution, which is defined as "the number of moles of solute dissolved in one litre of solution." Because volume is affected by temperature and ch
Similar Questions for you
After the 2023-24 revised CBSE syllabus, many chapters have been removed, and in some chapters syllabus is reduced. As per the latest CBSE exam pattern 2025-26, the Chapter 1 Solutions will be asked for 5-7 marks in the Class 12 Chemistry CBSE board exams.
For other competitive exams, the Solutions c
NCERT offers the foundation for every class 12 student. CBSE board exam questions are primarily based on the NCERT exercise, Examples and conceptual explanation.
There are various benefits of using NCERT Solutions.
- Specially for CBSE Board students, these NCERT Solutions are the best resource to
Shiksha focuses on completely fulfilling the needs of CBSE class 12 board students. We provide NCERT Solutions for classes 11th and 12th in class for physics, chemistry and mathematics.
Here are the key reasons why you should use Shiksha's NCERT Solutions for the Solution Chapter:
- Complete cover
If a solution containing more than 0.9% salt is used, and we place blood cells in it, the water will flow out of the cells, and they will shrink. Such a solution is a hypertonic solution.
Similarly, if a solution contains less than 0.9% salt, the water will flow into the cells, and they will enlarge.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 12th Chapter Two 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering