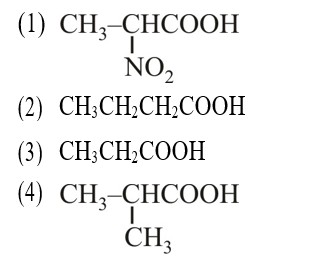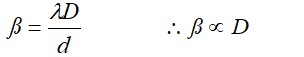Given below are two statements:
Statement I : The pH of rain water is normally
Statement II : If the pH of rain water drops below 5.6, it is called acid rain.
In the light of the above statements, choose the correct answer from the options given below
Given below are two statements:
Statement I : The pH of rain water is normally
Statement II : If the pH of rain water drops below 5.6, it is called acid rain.
In the light of the above statements, choose the correct answer from the options given below
Both statements are true when pH decreases from 5.6 then it becomes more acidic hence it is called Acid rain.
Similar Questions for you
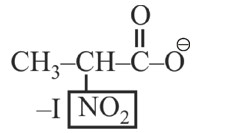
–I effect ∝ Acidic strength
+I effect ∝ Basic strength
* Most stable anion due to maximum –I effect.
* Most acidic
with increase in separation of screen from slits plane, fringe width increases.
Excessive nitrate in drinking water causes methemoglobinemia
Excessive nitrate in drinking water causes methemoglobinemia
Release of toxic/undesirable materials in the environment.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering