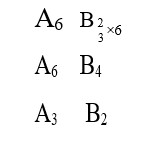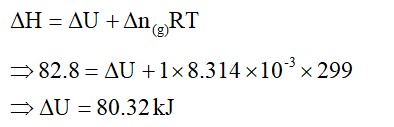Represent the potential energy/enthalpy change in the following processes graphically.
(a) Throwing a stone from the ground to roof.
(b) 12H2(g) + 12Cl2(g) HCl(g) ?rH? = –92.32 kJ/ mol
In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity?
Represent the potential energy/enthalpy change in the following processes graphically.
(a) Throwing a stone from the ground to roof.
(b) 12H2(g) + 12Cl2(g) HCl(g) ?rH? = –92.32 kJ/ mol
In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity?
This is a Short Answer Type Questions as classified in NCERT Exemplar
Throwing a stone from ground to roof
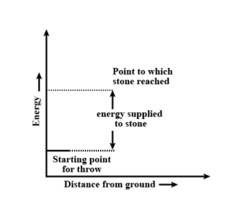
b) the reaction involved is a process where the energy decreases after the reaction. It can be represented as:In process b), potential energy/enthalpy change is a contributing factor to the spont
Similar Questions for you
Kindly go through the solution
(1) [Ni (NH3)6]+2 → Ni+2 → d8, C. No. = 6,
SP3d2, Para
(2) [Co (H2O)6]+2 → Co+2 → d6, C. No. = 6
d2sp3, Dia
(3) [Ti (H2O)6]+3 → Ti+3 → d1, C. No. = 6
d2SP3, Para
(4) [Co (NH3)6]+3 → Co+3 → d5, C. No. = 6
d2sp3, Para
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Six 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering