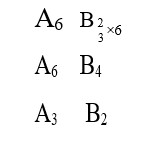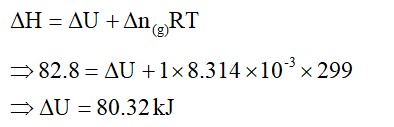Thermodynamics mainly deals with
(i) Interrelation of various forms of energy and their transformation from one form to another.
(ii) Energy changes in the processes which depend only on initial and final states of the microscopic systems containing a few molecules.
(iii) How and at what rate these energy transformations are carried out.
(iv) The system in equilibrium state or moving from one equilibrium state to another equilibrium state.
Thermodynamics mainly deals with
(i) Interrelation of various forms of energy and their transformation from one form to another.
(ii) Energy changes in the processes which depend only on initial and final states of the microscopic systems containing a few molecules.
(iii) How and at what rate these energy transformations are carried out.
(iv) The system in equilibrium state or moving from one equilibrium state to another equilibrium state.
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (i)
The laws of thermodynamics deal with energy changes of macroscopic systems involving a large number of molecules rather than microscopic systems containing a few molecules. Laws of thermodynamics apply only when a system is
Similar Questions for you
Kindly go through the solution
(1) [Ni (NH3)6]+2 → Ni+2 → d8, C. No. = 6,
SP3d2, Para
(2) [Co (H2O)6]+2 → Co+2 → d6, C. No. = 6
d2sp3, Dia
(3) [Ti (H2O)6]+3 → Ti+3 → d1, C. No. = 6
d2SP3, Para
(4) [Co (NH3)6]+3 → Co+3 → d5, C. No. = 6
d2sp3, Para
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Six 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering