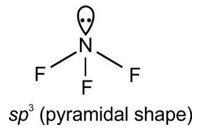
Which of the following statements are not correct?
(i) NaCl being an ionic compound is a good conductor of electricity in the solid state.
(ii) In canonical structures there is a difference in the arrangement of atoms.
(iii) Hybrid orbitals form stronger bonds than pure orbitals.
(iv) VSEPR Theory can explain the square planar geometry of XeF4.
Which of the following statements are not correct?
(i) NaCl being an ionic compound is a good conductor of electricity in the solid state.
(ii) In canonical structures there is a difference in the arrangement of atoms.
(iii) Hybrid orbitals form stronger bonds than pure orbitals.
(iv) VSEPR Theory can explain the square planar geometry of XeF4.
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and option (ii)
(i) NaCl being an ionic compound is a good conductor of electricity in the molten state. It is not a bad conductor of electricity in solid state. So, the statement mentioned in the question is not corr
Similar Questions for you
He2 has zero bond order hence it does not exist.
The three fundamental laws of chemistry are - Law of Definite Proportions, Law of Conservation of Mass, and Law of Multiple Proportions.
The three types of chemical bonds are - ionic, metallic and covalent bonds. When the electrons transfer between the atoms, they form the Ionic bonds by producing charged ions that are attracted to each other. When atoms share electrons, covalent bonds are created. When metal atoms share a sea of del
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Four 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering