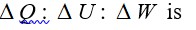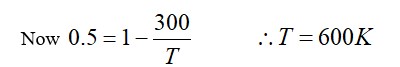A heat engine is involved with exchange of heat of 1915 J, –40 J, +125 J and –QJ, during one cycle achieving and efficiency of 50.0%. The value of Q is:
A heat engine is involved with exchange of heat of 1915 J, –40 J, +125 J and –QJ, during one cycle achieving and efficiency of 50.0%. The value of Q is:
Option 1 - <p>40 J<br><!-- [if !supportLineBreakNewLine]--><br><!--[endif]--></p>
Option 2 - <p>640 J</p>
Option 3 - <p>400 J</p>
Option 4 - <p>980 J</p>
7 Views|Posted 9 months ago
Asked by Shiksha User
1 Answer
A
Answered by
9 months ago
Correct Option - 4
Detailed Solution:
Efficiency η = Q_net/Q? = (1915-40+125-Q)/ (1915+125) = 0.5.
(2000-Q)/2040=0.5 ⇒ Q=980J.
Similar Questions for you
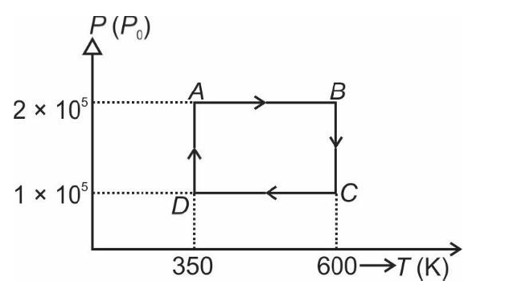
From A to B the process is isobaric
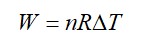
= W = 2 × R (600 - 350)
= 500 R
Heat is path dependent so path function but internal energy does not depend on path chosen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
7L
Reviews
|
1.9M
Answers
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering