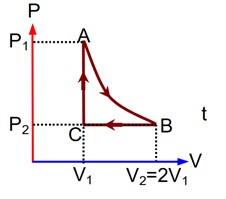
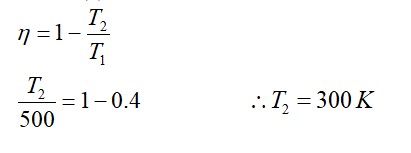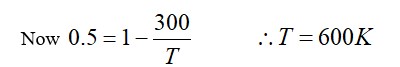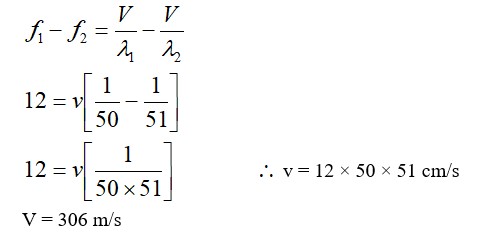n mole of perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes.
A->B : Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 and Pressure changes from P1 to P2.
B->C : Isobaric compression at pressure P2 to initial volume V1.
C->A : Isobaric change leading to change of pressure from P2 to P1.
Total work done in the complete cycle ABCA is:
n mole of perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes.
A->B : Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 and Pressure changes from P1 to P2.
B->C : Isobaric compression at pressure P2 to initial volume V1.
C->A : Isobaric change leading to change of pressure from P2 to P1.
Total work done in the complete cycle ABCA is:
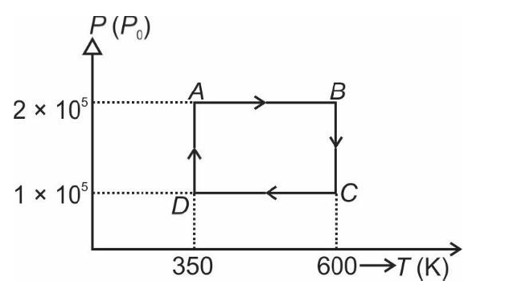
[At B, 2P2 V1 = nRT]
Similar Questions for you
From A to B the process is isobaric
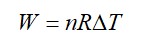
= W = 2 × R (600 - 350)
= 500 R
Heat is path dependent so path function but internal energy does not depend on path chosen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering