Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) Lv=540 kcal kg-1
= 540 kg-1 = 540 4.2jkg-1
Energy required to evaporate 1kg of water = Lv kcal
And MA kg of water requires MALV kcal
Since there are NA molecules in MA kg of water the energy required for 1 molecule to evaporate
Is
U=
=
=90
= 6.8
(b) Let the water molecules to be points and are separated at a distance d from each other
volume of NA molecule of water =
thus the volume of one molecule is =
the volume around one molecule is d3=
d=
d= 3.1
(c) 1 kg of vapour occupies volume =1601 m3
18 kg of vapour occupies 18 m3
6 molecules occupies 18 m3
1 mo
New Question
10 months agoNew Question
10 months agoContributor-Level 10
To participate in the admission process NIMS - Nehru Institute of Management Studies courses, students must receive a minimum score of 50% aggregate in their graduation. This academic barrier aids in the selection of applicants with a strong educational background and foundational knowledge in the management sector. The grade demonstrates that students have performed well in the classroom and are prepared for the challenging portion of management education.
New Question
10 months agoNew Question
10 months agoContributor-Level 6
For CBSE 10th Maths, one must focus on NCERT book. It includes all the topics mentioned in the CBSE 10th Maths syllabus and covers them thoroughly so as to clear the concepts. After finishing the NCERT textbook, if you wish to refer other reference books for practising more problems, you can refer R.D. Sharma and R.S. Aggarwal.
Here's a more detailed breakdown-
The NCERT textbook is the primary source for learning and understanding concepts. It covers all the topics of the syllabus.
RD Sharma offers detailed solutions and provides numerous pratice problems which are very useful in clearing doubts
RS Aggarwal book offers a wide
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) consider a horizontal parcel of air with cross section A and height dh
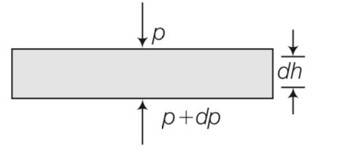
Let the pressure on the top surface and bottom surface be P and p+dp. If the parcel is in equilibrium , then the net upward force must be balanced by the weight
(P+dP)-PA=-
dP= -
negative sign shows that pressure decreases with height.
(b) let o be the density of air on the surface of earth.
As per question , pressure density
dP= -
In
P=Poe(- )
(c) as P =Po
in
p=1/10 Po
in( ) =-
in1/10 =-
h=- in1/10= - -1=
=
=
= 16 103m
(d) we know that
P , temperature remain
New Question
10 months agoNew Question
10 months agoContributor-Level 9
Of course, a Bachelor of Mass Media is an undergraduate degree programme that will provide you with the necessary knowledge, skills, and networking opportunities to thrive in the media industry. It will give you learning, different career options, industry connections, and more.
New Question
10 months agoContributor-Level 9
It totally depends on your interests and career goals. Some popular MBA specializations you can follow after BMM are
- MBA in Marketing: If you're into advertising, brand management, digital marketing, etc.
- MBA in International Business: If you wish to explore the global markets related to media and communication.
- MBA in Media Management: If the business idea of media companies, broadcasting, publishing, etc., excites you.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
The M.A. in Tourism Management programme offered by the Department of Continuing Education and Extension (DCEE) at Delhi University is a new, industry-driven programme designed to equip students with both theoretical knowledge and practical skills for careers in the tourism sector. It aims to provide a comprehensive understanding of tourism management, including heritage tourism, sustainable tourism, tourism marketing, and digital tourism platforms. The programme emphasizes application-based learning, live projects, and industry exposure, preparing graduates for opportunities in both national and international tourism, hospitality, and
New Question
10 months agoContributor-Level 10
The acceptance rate of Sacred Heart University is around 68% over the years, which means out of 100 applicants, around 68 are selected. Hence, it can be said that the university is moderately selective and the admissions are fairly competitive. Applicants can increase their chances of acceptance by meeting the admission requirements and carefully submitting the required documents as per the programs chosen by the applicant.
New Question
10 months agoContributor-Level 10
The Sacred Heart University offers programs for applicants all over the world to choose, but some of them are more popular than others. the university offers more than 67 courses for international students in both UG and PG levels. Some of the popular majors are mentioned below:
- Health Professions and Related Programs
- Business, Management, Marketing
- Psychology
- Biological and Biomedical Sciences
- Communication, Journalism, and Related Programs
- Law Enforcement
- Social Sciences
- Visual and Performing Arts
- English Language and Literature/Letters.
New Question
10 months agoContributor-Level 9
New Question
10 months agoContributor-Level 9
All courses available at CMR Institute of Technology are filled by entering through an entrance test. If students don't come from Karnataka, then admission may take place through the Management Admission Process based on their ranking in merit. Candidates can get direct admission using this process. Candidates have to meet some basic requirements to be eligible.
New Question
10 months agoContributor-Level 9
Requirements for admission through KCET are a high score in Class 12 and a good rank in the KCET exam which is not specified in general. Cuts may change depending on the engineering branch selected. When you have a higher rank, your chance of getting chosen for your course becomes better. Students from the reserved category in Karnataka have reduced cutoff criteria.
New Question
10 months agoContributor-Level 10
Yes, admission are open at CMR Institute of Technology Bengaluru. The institute accepts admisison based on both merit and entrance-based for Karnataka and non-Karnataka students. The university conducts CMRUAT for shortlisting aspirants for specific courses. In addition to this, the university also accepts national or state-level entrance exams such as JEE Main, COMEDK UGET, etc.
New Question
10 months agoContributor-Level 10
Shikshapeeth College of Management and Technology BCA the highest package for the year 2025 is yet to be released. However, as per the previous placements records, the highest and average package offered during Shikshapeeth College of Management and Technology placements 2023 stood at INR 6.50 LPA and INR 4.50 LPA, respectively.
New Question
10 months agoContributor-Level 10
To get a seat in the BCA course at Shikshapeeth College of Management and Technology, candidates must complete their Class 12/ entrance exam with decent scores. Further, they are required to fill out the application form online/offline. The college shortlists the candidates on the basis of their scores secured in their Class 12 or equivalent, and shortlisted candidates will be called for PI/ Group discussion.
New Question
10 months agoContributor-Level 10
The total number of seat intakes for the BCA course at Shikshapeeth College of Management and Technology is 120. To get a seat in this course, candidates are required to perform well in their Class 12/ entrance exam and in the group discussion/ PI conducted by the admission authority of the college. Check out the number of seats allocated to the course in the table below:
| Courses | Seats |
|---|---|
| Bachelor of Computer Application (BCA) | 120 |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
