Ask & Answer: India's Largest Education Community
All Questions
New Question
9 months agoContributor-Level 10
Given are the oxide of alkali and alkaline earth metals which are ionic in nature.
Simple oxide are Li2O, CaO, MgO and K2O.
Peroxide is Na2O2 and superoxide is KO2.
All simple oxides are diamagnetic as it has no unpaired electron.
New Question
9 months agoContributor-Level 10
Yes, Germany is a fantastic place to earn a Master's degree. Germany boasts a top-notch educational system and several institutions that are famous among students from across the globe.
Moreover, Germany is a desirable option for overseas students since it provides low or even free tuition, as well as research opportunities, work experience, and the ability to study abroad and experience a different culture are all available to students at German institutions. Overall, for students seeking a top-notch education and a worldwide perspective on their subject of study, earning an MS in Germany might be a great choice.
New Question
9 months agoContributor-Level 10
BSc programme at Ajeenkya DY Patil University Seamedu has a Shiksha Rating of 3.6/5, which is higher than Savitribai Phule Pune University (3.0/5) but lower than MIT-WPU (4.3/5). This suggests moderate student satisfaction in terms of academics, faculty, and infrastructure.
New Question
9 months agoContributor-Level 10
BSc course fees at Kumaraguru College of Liberal Arts and Science has not been revealed for the year 2025. Other BSc institutes similar to Kumaraguru College of Liberal Arts and Science have a BSc tuition fee range between INR 4 lakh- 4.5 lakh. Candidates who wish to enrol in BSc courses at Kumaraguru College of Liberal Arts and Science should keep checking the official website for any updates.
New Question
9 months agoNew Question
9 months agoContributor-Level 10
Effective number of atom in C.C.P
= 4
Number of octahedral void = 4
Number of cations = 4
Number of anion = 4
Formula of compound = A4B4
Empirical formula = AB
Ans. x = 1
New Question
9 months agoContributor-Level 10
50 ml of 1 (M) HCl + 30 ml of 1 (M) NaOH
NaOH + HCl -> NaCl + H2O
30 × 1 mmol 50 × 1 mmol
0 mmol 20 mmol
x = 6021
Ans. = 6021
New Question
9 months agoContributor-Level 10
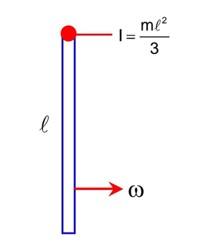
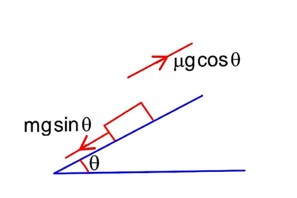
By work energy theorem
Work done = change in K.E.
Work done by friction work done by spring
As 90% of K.E. is losed by friction so that
-K -> -16 × 105
K = 16 × 105
New Question
9 months agoContributor-Level 10
The NBEMS will release the FMGE 2026 results on the official website. According to the notification, the FMGE result will be out in the last week of August 2026, for the June session and the last week of January. 2027, for the December session exam.
New Question
9 months agoContributor-Level 10
In the first year of BSc at Ajeenkya DY Patil University, students build a strong foundation in both creative and technical aspects of game development. They begin with Visual Programming Basics, Programming in C#, and Game Design-I, while also learning to navigate game engines. Subjects like Communication Skills and Environmental Science ensure a holistic start.
New Question
9 months agoContributor-Level 10
Jayoti Vidyapeeth Women's University offers the flagship BTech programme in a variety of specializations. The course is a popular programme for students seeking to build a career in Engineering. The total tuition fees for Jayoti Vidyapeeth Women's University BTech is INR 4 L - 5.5 L. Jayoti Vidyapeeth Women's University BTech admissions are conducted on the basis of merit.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts