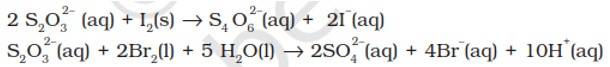Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
5.23. (a) The dipole-dipole interaction between two HCl molecules is stronger than the London forces but is weaker than ion-ion interaction because only partial charges are involved.
New Question
11 months agoContributor-Level 10
Reactions (a) and (b) indicate that H3PO2 (hypophosphorous acid) is a reducing agent and thus reduces both AgNO3 and CuSO4 to Ag and Cu respectively. Conversely, both AgNO3 and CuSO4 act as oxidising agent and thus oxidise H3PO2to H3PO4 (orthophosphoric acid) Reaction (c) suggests that [Ag (NH3)2]+ oxidises C6H5CHO (benzaldehyde) to C6H5COO– (benzoate ion) but reaction (d) indicates that Cu2+ ions cannot oxidise C6H5CHO to C6H5COO–. Therefore, from the above reactions, we conclude that Ag+ ion is a strong deoxidising agent than Cu2+ ion.
New Question
11 months agoContributor-Level 10
Department of Management Studies applications are open for MBA course. Candidates can apply online on the university's website for admission.
New Question
11 months agoContributor-Level 10
XeO64−? oxidizes F− and F− reduces XeO64−?
Hence, the given reaction occurs.
The oxidation number of Xe decreases from +8 to +6. The oxidation number of F increases from -1 to 0.
Thus, Na4? XeO6? is a stronger oxidising agent than F−.
New Question
11 months agoContributor-Level 10
During admission season, students often get confused about college selection. Cluster University Srinagar BBA/ BBA (H) can be compared based on various parameters. The students can compare preferred colleges/ universities based on a list of parameters. A few parameters that can be considered for college comparison are fees, reputation, faculty, placements, etc.. However, the importance of a parameter can vary from one student to another.
New Question
11 months agoContributor-Level 10
Yes, there are many government Paramedical colleges in India. Some of them are mentioned below along with their tuition fees:
Public/Govt. Colleges | Tuition Fee |
|---|---|
AIIMS Delhi Admission | INR 1,000 – INR 3,000 |
Jamia Hamdard Admission | INR 90,000 – INR 6 lakh |
JIPMER Puducherry Admission | INR 2,000 – INR 47,000 |
AIIMS Rishikesh Admission | INR 650 – INR 2 lakh |
NIMHANS Admission | INR 30,000 – INR 2 lakh |
Disclaimer: This info. is taken from official website and may vary.
New Question
11 months agoGuide-Level 15
Sobhasaria Group of Institutions offers admission to MBA programme based on CMAT scores of the candidates. Selected candidates have to further participate in the Online Counselling conducted by the Convener, RMAP. The institute offers admission on a total of 210 seats of the MBA programme.
New Question
11 months agoContributor-Level 10
Fluorine oxidizes chloride ion to chlorine, bromide ion to bromine and iodide ion to iodine respectively.
F2? + 2Cl− → 2F− + Cl2?
F2? + 2Br− → 2F− + Br2?
F2? + 2I− → 2F− + I2?
Chlorine oxidizes bromide ion to bromine and iodide ion to iodine.
Cl2? + Br− → 2Cl− + Br2?
Cl2? + I− → 2Cl− + I2?
Bromine oxidizes iodide ion to iodine.
Br2? + I− → 2Br− + I2?
But bromine and chlorine cannot oxidize fluoride to fluorine. Hence, fluorine is the best oxidizing agent amongst the halogens. The decreasing order of the oxidizing power of halogens is&nb
New Question
11 months agoContributor-Level 10
Yes, there are many private Paramedical colleges in India. Some of them are mentioned below along with their tuition fees:
Private Colleges | Tuition Fee |
|---|---|
CMC Vellore Admission | INR 400 – INR 2,400 |
JSS College of Pharmacy Admission | INR 40,000 |
KMC Admission | INR 3 Lacs – INR 37 lakh |
Dr. DY Patil Vidyapeeth, Pune Admission | INR 2 Lacs – INR 6 lakh |
Saveetha Institute of Medical and Technical Sciences Admission | INR 2 Lacs – INR 13 lakh |
Disclaimer: This information is sourced from official website and may vary.
New Question
11 months agoContributor-Level 10
Yes, it is estimated that joining the best Paramedical colleges in India can be affordable as most colleges have tuition fees of less than INR 1 lakh. Listed below are some colleges along with their affordable fees:
Low-cost Colleges | Tuition Fee |
|---|---|
AIIMS Delhi | INR 1,000 – INR 3,000 |
CMC Vellore | INR 400 – INR 2,000 |
PGIMER Chandigarh | INR 7,000 |
Jamia Hamdard | INR 90,000 |
Disclaimer: This info. is taken from the official website and may vary.
New Question
11 months agoContributor-Level 10
The SRM University BSc fee structure is a composition of various fee elements to be paid by the students to complete the course. According to the fee structure, students are required to pay three major components: tuition fees, hostel fees, and a one-time fee.
Moreover, students must note that only the ones who opt for hostel facilities are charged a hostel fee. Combining all, the total fees for the BSc programme ranges from INR 7.65 Lacs to INR 10.15 lakh. Check the below table to learn the component-wise bifurcation of the total fees:
Fee Components | Amount |
|---|---|
Tuition Fees | INR 4.65 Lacs - INR 6.2 lakh |
Hostel Fees | INR 2.85 Lacs - INR 3.8 lakh |
One-time Payment | INR 15,000 |
Total Fees | INR 7.65 Lacs - INR 10.15 lakh |
Note: The above-mentioned fee is as per the official sources. However, it is indic
New Question
11 months agoContributor-Level 10
The average O.N. of S in S2O32- is +2 while in S4O62- it is + 2.5. The O.N. of S in SO42- is +6. Since Br2 is a stronger oxidising agent than I2, it oxidises S of S2O32- to a higher oxidation state of +6 and hence forms SO42- ion. I2, however, being weaker oxidising agent oxidises S of S2O32- ion to a lower oxidation of +2.5 in S4O62- ion. It is because of this reason that thiosulphate reacts differently with Br2 and I2.
New Question
11 months agoContributor-Level 10
There are various positive aspects as to why students should choose to study MSc Forensic Science courses.
- Contribute to Justice: Forensic Science courses play a crucial role in the criminal justice system by helping to identify, apprehend, and convict criminals while also exonerating the innocent. By studying Forensic Science courses, candidates can directly contribute to ensuring a fair and just legal process.
- Varied Career Opportunities: Graduates of Forensic Science courses can work in a variety of settings, including crime laboratories, law enforcement agencies, medical examiner's offices, research institutions, and privat
New Question
11 months agoContributor-Level 10
Joining the top Paramedical colleges in India can be worthwhile for you. For that, you need to check its ROI. Listed below are some colleges along with their tuition fees and average placement package:
Top Colleges | Cost | Placement Package |
|---|---|---|
AIMMS Delhi Course | INR 1,000 – INR 3,000 | INR 12 LPA – INR 24 LPA |
CMC Vellore Courses | INR 400 – INR 2,000 | INR 6 LPA – INR 8 LPA |
PGIMER Chandigarh Courses | INR 7,000 – INR 20 lakh | INR 9 LPA – INR 18 LPA |
Jamia Hamdard Courses | INR 90,000 – INR 6 lakh | INR 3 LPA – INR 6 LPA |
JIPMER Puducherry Courses | INR 2,000 – INR 47,000 | INR 6 LPA – INR 24 LPA |
Disclaimer: This information is sourced from official website/ media reports/ NIRF website and may vary.
New Question
11 months agoContributor-Level 10
Substance oxidised | Substance reduced | Oxidising agent | Reducing agent |
(a) C6H12O6 | AgBr | C6H12O6 | |
(b) HCHO | [Ag (NH3)2]+ | [Ag (NH3)2]+ | HCHO |
(c) HCHO | Cu2+ | HCHO | |
(d) N2H4 | H2O2 | N2H4 | |
(e) Pb | PbO2 | Pb |
New Question
11 months agoContributor-Level 10
11.86

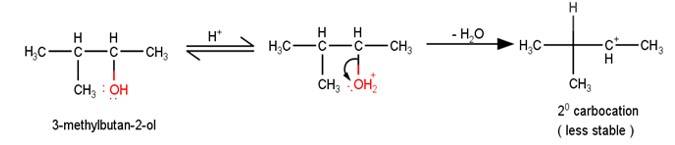
The first step in the mechanism of th e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
2. The next step is a rearrangement of the 20 carbocations formed in the above step is less stable it rearranges by a 1,2-hydride shift to form more stable 3° carbocations.
New Question
11 months agoContributor-Level 10
The general eligibility criteria for admission to the best Paramedical colleges in India are mentioned below:
- UG: Candidates should have completed their Class 12 or equivalent with a Science background (Physics, Chemistry, Biology) with a passing percentage of 50%.
- PG: Candidates typically need a relevant bachelor's degree with a passing aggregate of 50% in a related field of study.
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoGuide-Level 15
The total semester fee for MBA at Sobhasaria Group of Institutions is INR 1.60 lakh. Candidates need to pay INR 40,000 semester-wise for MBA. Aspirants also need to pay a one-time refundable caution deposit of INR 5,000. Besides, candidates also have to pay a registration fee of INR 2,500 for the MBA course.
New Question
11 months agoContributor-Level 10
(a) Toluene can be oxidised to benzoic acid in acidic, basic and neutral media according to the following redox equations:
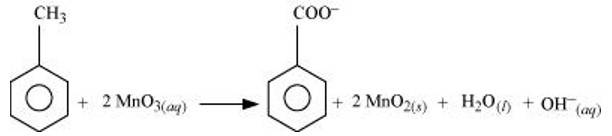
In the laboratory, benzoic acid is usually prepared by alcoholic KMnO4 oxidation of toluene. However, in industry alcoholic KMnO4 is preferred over acidic or alkaline KMnO4 because of the following reasons:
(i) The cost of adding an acid or the base is avoided because in the neutral medium, the base (OH- ions) are produced in the reaction itself.
(ii) Since reactions occur faster in homogeneous medium than in heterogeneous medium, therefore, alcohol helps in mixing the two reactants, i.e., KMnO4
New Question
11 months agoContributor-Level 9
Out of the total 145 seats, the majority are at the Abdul Ahad Azad Memorial College affiliated with Cluster University Srinagar. The college has 100 seats for the UG-level course. The given seat details are as per the official website/ sanctioning body. It is still subject to changes and hence, is indicative.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts