Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
NO3− + 3Fe2+ + 4H+→ NO+ + 3Fe3+ + 2H2O
[Fe (H2O)6]2+ + NO → [Fe (H2O)5 (NO)]2+ + H2O ( brown ring )
New Question
10 months agoContributor-Level 10
Students are advised to correct the errors even if it as a minor mistake. They must inform the school authorities about the error in the Odisha BSE hall ticket. School authorities will help you in getting the new hall ticket without any mistakes.
New Question
10 months agoContributor-Level 6
TS SSC supplementary result 2026 will be released in June on the official website. Students can check their TS SSC supply result using roll number and date of birth.
New Question
10 months agoContributor-Level 10
It is mandatory to have 75% attendance to get hall ticket for the board examinations. However, you may submit a medical certificate in case you were not well or due to any other medical condition to get a waiver.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
A is PCl5 (yellowish white powder)
P4 + 10Cl2→4PCl5
B is PCl3 (colourless oily liquid)
Hydrolysis of the following product
PCl3+3H2O→H3PO3+3HCl
PCl5+4H2O→H3PO4+5HCl
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Six F atoms can be accommodated around sulphur due to fluorine's tiny size, however the chloride ion is comparably bigger, resulting in interatomic repulsion.
New Question
10 months agoContributor-Level 10
Following are some tips to improve the writing and presentation skills for the Odisha BSE board exams.
- Students must write the answers neatly. The hand writing should be legible.
- Write the answers with proper heading and subheading.
- Highlight your important points.
- Cross out the errors neatly with single line.
- Divide the time for each question to avoid rushing.
- Practice writing the previous year question papers.
New Question
10 months agoContributor-Level 10
The fee for the courses at Renaissance College of Commerce and Management are as follows:
| Courses | Tuition Fees |
|---|---|
B.Com (3 courses) | INR 75 K - 1.2 lakh |
BBA (2 courses) | INR 1.9 L - 2.6 lakh |
B.A. (1 course) | INR 1.1 lakh |
Candidates who receive the final admission offer are eligible to pay the course fee and secure their seat.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Oxygen is more electronegative than sulphur, the bond angle of H2O is greater, and the bod pair electron of an OH bond will be closer to oxygen, causing bond-pair repulsion between bond pairs of two OH bonds
New Question
10 months agoContributor-Level 10
The Board of Secondary Education Odisha, will not release the list of BSE Odisha Class 10th toppers 2025. Odisha did not release the list of BSE Odisha Class 10th toppers 2024. Instead, the board released list of top performing districts.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Chlorine has vacant d-orbitals, which become excited upon bonding when electrons from the 3p-orbital are promoted to the 3d-orbital, giving it a covalency of three.
Due to the lack of unoccupied d-orbitals in the second energy shell, fluorine cannot expand its octet. As a result, it can only have one covalency.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
NO2 exists as a monomer with one unpaired electron in the gaseous state, but it dimerises to N2O4 in the solid state, leaving no unpaired electron, making the solid form diamagnetic.
New Question
10 months agoContributor-Level 10
The course duration for the BCom programme at Renaissance College of Commerce and Management is three years. BCom at RCCM develops proficiency in a specific field, such as business economics, financial accounting, cost accounting, income tax, auditing, business finance, business law, marketing, and business communication. The course focuses on giving candidates a wide range of managerial abilities as well.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
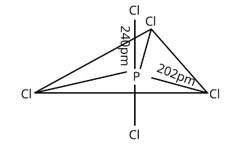
Three P—Cl bonds are arranged in one plane at a 1200 angle. These bonds are known as equatorial bonds because they are formed between two points. The remaining two P—Cl bonds, one above the equatorial plane and the other below it, form 90° angle with the plane. Axial bonds are the name for these types of bonds. Because axial bond pairs are subjected to greater repulsive interaction than equatorial bond pairs, axial bonds are slightly longer and hence slightly weaker than equatorial bonds, making the PCl5 molecule more
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
PH3 is insoluble in water and cannot create hydrogen bonds with it, it forms bubbles, whereas NH3 dissolves because it is soluble in water and can form hydrogen bonds with it.
New Question
10 months agoContributor-Level 9
Having scored 600 out of 1000 in CUET 2025, there is a good chance of getting into mid-tier central and state universities for a BA program. You may find it challenging to get into top colleges like Hindu College or Miranda House of Delhi University, since colleges like Ramjas, Zakir Husain, and colleges under BHU and AMU may be a bit easier to get into, depending on your category and subject combination. Keep an eye on the cutoffs and apply broadly, in addition to colleges that are accepting CUET scores, think about state universities and private colleges too. The probability for you as an applicant will improve if you are flexible in
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
New Question
10 months agoContributor-Level 10
KK University offers various courses at UG and PG levels. Before finalising the university for admission, candidates must compare it with other options to make a suitable choice. The factors that can be used to compare two or more universities depend on the candidates' preferences and priorities. Some factors that can be used while comparing KK University with other universities or institutes offering similar courses are listed below:
- Fees, placements, ROI
- Location, Infrastructure and facilities
- Course structure and curriculum
- Medium of instruction
- Eligibility & Selection Criteria
- Rankings and accreditations
- Student reviews on all the a
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
4NH3 + 5O2 ![]() 4NO + 6H2O
4NO + 6H2O
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts