Ask & Answer: India's Largest Education Community
All Questions
New Question
8 months agoContributor-Level 10
Bohr’s model played an important role in the development of quantum theory. It introduced the idea of quantised electron orbits. It disproved claims of classical mechanics, which predicted that electrons would spiral into the nucleus. Bohr proposed that electrons can exist only in specific, stable energy levels called stationary states. There would be transitions between these levels that would help Bohr explain the line spectra of hydrogen. That together linked the atomic structure with the concept of energy quantisation. Even though the model could not explain multi-electron atoms, it laid the foundation for modern quantum mech
New Question
8 months agoContributor-Level 10
Metal of group 7, 8, & 9 dose not form interstitial hydride this is called hydride gap.
Mn → group - 7
Fe → group - 8
Co → group - 9
So, Cr will forms interstitial hydride.
New Question
8 months agoContributor-Level 10
For admission to the IHM Hajipur BSc course, applicants must meet the minimum eligibility criteria set by the institution for their desired course. The table below lists the IHM Hajipur courses along with their corresponding eligibility criteria:
| Courses | Eligibility | Selection |
|---|---|---|
BSc | Class 12 | NCHMCT JEE |
New Question
8 months agoContributor-Level 10
Conjugate base is highly stable.
Acidic strength ∝ stability of conjugate base.
So, 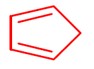
New Question
8 months agoContributor-Level 10
Yes, IHM Hajipur does offer a BSc course for the duration of three years, divided into six semesters. Further, it is offered in full-time mode. The minimum eligibility requirement to get admission into the BSc course at IHM Hajipur is that candidates must complete their Class 12 with English as one of the subjects.
New Question
8 months agoContributor-Level 10
The existence of atomic spectra tells us that energy levels in atoms are quantised. When atoms absorb or emit light, they do so at specific wavelengths. They lead to line spectra instead of a continuous spectrum. Now, every line corresponds to an electron that transitions between fixed energy levels. This is to make the photon's energy equal to the difference between them. If energy levels were not discrete, the spectra would be continuous. So, the line spectra provide direct evidence that electrons in atoms occupy quantised energy states.
New Question
8 months agoContributor-Level 10
Phase | Medium | Colloid Type |
| (1) Cheese | liquid | solid | Gel |
| (2) Pumice stone | gas | solid | Solid sol |
| (3) Hair cream | liquid | liquid | Emulsion |
| (4) Cloud | liquid | gas | Aerosol |
New Question
8 months agoContributor-Level 10
O? = σ1s² σ1s² σ2s² σ2s² σ2pz² π2px² = π2py² π2px¹ = π2py¹
O? = σ1s² σ1s² σ2s² σ2s² σ2pz² π2px² = π2py² π2px¹
O? = σ1s² σ1s² σ2s² σ2s² σ2pz² π2px² = π2py² π2px² = π2py¹
O? ²? = σ1s² σ1s² σ2s² σ2s² σ2pz² π2px² = π2py² π2px² = π*2py²
B.O = (Bonding e? - Antibonding e? )/2
B.O of O? = (10 - 6)/2 = 2
B.O of
New Question
8 months agoContributor-Level 10
Li? CO? decomposes easily on heating as;
Li? CO? - (Δ)-> Li? O + CO? ↑
NaHCO? is used in dry fire extinguishers.
K is most abundant element in cell fluid.
CsI is least soluble due to smaller hydration energy of Cs? & I?
New Question
8 months agoContributor-Level 10
Chelation increases stability of complex. Also more the chelation more the stability. Here, [Co (en)? (NH? )? ]Cl? , [Co (en)? ]Cl? and [Co (en) (NH? )? ]Cl? have chelation due to ethylenediamine. Therefore [Co (en)? ]Cl? has the highest chelation so the highest stability.
New Question
8 months agoContributor-Level 10
Using, µ = √n (n+2) B.M, n = number of unpaired electrons.
? Ti? ³ = 4s? 3d¹, unpaired electron = 1
µ = 1.73 B.M
? V? ² = 4s? 3d³, unpaired electron = 3
µ = 3.87 B.M
? Sc? ³ = 4s? 3d? , unpaired electron = 0
µ = 0 B.M
New Question
8 months agoContributor-Level 10
Anions have larger radii than atoms. Also, higher the e/p ratio higher the ionic radii. So, N? ³ > O? ² > F?
New Question
8 months agoContributor-Level 10
BSc Computer Science core subjects are mentioned in the table below:
BSc Computer Science Core Subjects | |
| Programming in C | Linux Operating System |
| Fundamentals of Information Technology | SQL AND PL/SQL |
| Object Oriented Programming using 'C+' | Compiler Construction |
| System Analysis and Design | Database Management System |
| java Programming | Visual Basic Programming |
| Operating Systems | Robotics & Perception |
Note: The above information is sourced from external sites and may vary
New Question
8 months agoContributor-Level 10
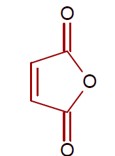
Heating of cis-but-2-enedioic acid gives maleic anhydride as shown.
[Chemical reaction showing conversion of cis-but-2-enedioic acid to maleic anhydride upon heating]
New Question
8 months agoContributor-Level 10
CFC breakdown by visible light to give Cl radical which react with stratospheric ozone.
CFC, CF? Cl? - (hv)-> Cl• + •CF? Cl
Cl• (g) + O? → ClO• + O?
ClO• + O → Cl• + O?
Atmospheric ozone reacts with NO to give NO? and O?
O? + NO → NO? + O?
New Question
8 months agoContributor-Level 10
Admission decisions at SNU are based on a number of factors, such as academic achievement like GPA, personal statements, letters of recommendation, study plan, as well as other equivalent records and / or descriptions of specific qualifications. The admissions committee considers the level of studies completed, the quality of achievement, aptness of goals, suitability for the preparation of the proposed programme of studies, and characteristics of the last institution attended. Also, proficiency in Korean and other languages are also considered.
New Question
8 months agoContributor-Level 10
SNU Global Scholarship falls in the category of scholarship for international students in enrolled in Graduate programs. The number of recipients of this university is 160 approximately It covers the tuition fee, living expenses, housing cost, and airfare. Also, language support can be offered for those who seek to master Korean language. Additional funding can be offered to those who require academic support.
New Question
8 months agoContributor-Level 10
SNU South Korea is a public research university in Seoul, South Korea. As a result of the university's commitment to fostering productive research environment, many of this university's departments and research institutes, especially in Engineering and Natural Sciences fields are globally recognised for its cutting-edge research. They are placed within the Top 20 in terms of citations and publications of the Scientific Citation Index.
New Question
8 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts