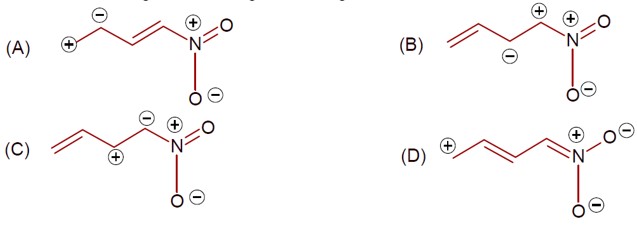Ask & Answer: India's Largest Education Community
All Questions
New Question
8 months agoContributor-Level 10
Organic compound + Cl? → A (Containing Cl)
5gm
A + AgNO? → AgCl
0.5 gm → 0.3849gm
Mole of AgCl = 0.3849 / 143.32
Mass of Cl = (0.3849/143.32) × 35.5 ≈ 0.095
% of Cl in A = (0.095/0.5) × 100 = 19%
New Question
8 months agoContributor-Level 9
Oxidation states:
CrO? (+6), Fe?O? (+3), MnO? (+4), V?O? (+5), Cu?O (+1)
(a), (b), (c), (d), (e)
Order of oxidation numbers:
Cu?O < Fe?O? < MnO? < V?O? < CrO?. Hence correct order is; e < b < c < d < a
New Question
8 months agoContributor-Level 10
In triethyl amine, nitrogen is sp³ hybridized, hence bond angle is approximately 109°28' but since lone pair- bond pair repulsion is greater than bp-bp repulsion. Hence, the exact angle is found to be 108°.
New Question
8 months agoContributor-Level 10
Number of GI in [Co (NH? )? (NO? )? ] is 2 i.e, X
Number of GI in [Cr (C? O? )? ]³? is 0 i.e. Y
[X + Y] = 2
New Question
8 months agoContributor-Level 10
The entrance exam scores accepted by IHM Hajipur are from the National Council for Hotel Management & Catering Technology Joint Entrance Examination, i.e., NCHMCT JEE MSc. It's a national-level exam conducted by the NCHMCT for admissions to MSc programmes in various IHM institutes across India. To apply for this entrance exam, candidates are obliged to fulfil the minimum eligibility requirements first.
New Question
8 months agoContributor-Level 10
Λ? = K × 1000 / M = (2 × 10? × 10³) / 10? ³ = 20 Scm²mole? ¹
For weak acid (α) = Λ? / Λ? = 20 / 190 = 2/19
K? = Cα² / (1 - α) = 10? ³ × (2/19)² / (1 - 2/19)
= 10? ³ × (4/361) / (17/19) = 0.01238 × 10? ³
= 12.38 × 10?
So, ans is 12 (the nearest integer).
New Question
8 months agoContributor-Level 10
Pearl Academy, Jaipur offers the major degree course of Bachelor of Design (BDes) its which is a widely chosen pattern among the students of Design. The overall tuition expenses for the BDes course at Pearl Academy is around INR 20.42 L - 26.4 L. The Institute offers this course in different areas of specialisation. Students are allowed to select the specialisation that matches their professional goals the best. Different Pearl Academy, Jaipur BDes specialisations are given as follows: B.Des. in Interior Architecture Design B.Des. in Fashion Design B.Des. in Communication Design
New Question
8 months agoContributor-Level 10
Joining MBA course at Excel Engineering College is generally considered a decent option. However, the "good" factor depends on various factors such as infrastructure, faculty experience, hostel and mess facilities, curriculum, etc. This can vary from candidate to candidate.
New Question
8 months agoContributor-Level 10
Adarsh Group of Institutions offers Master of Buisness Administration (MBA) programme through AIMIT. The course is offered in various specialisations mainly in the areas of Finance, Marketing, Human Resource, Business Analytics, etc. The course duration is two years, spread across four semesters.
New Question
8 months agoContributor-Level 10
In Aniline, lone pair is delocalized on less EN carbon atom while in acetamide it is delocalized on more EN oxygen atom. Hence aniline is more basic than acetamide.
New Question
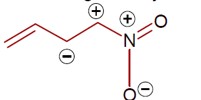
8 months agoContributor-Level 9
Same charge on adjacent atom is not stable. Hence the incorrect resonating structure is,
[Image of the incorrect resonance structure with adjacent positive charges]
New Question
8 months agoContributor-Level 10
The MBA course at Excel Engineering College is offered for a duration of two years. Candidates willing to take admission in this course must appear for the TANCET exam and achieve a valid aggregate in the same. Candidates who have passed graduation with a minimum of 45% aggregate (40% for reserved category) are eligible for admission in the MBA course at Excel Engineering College.
New Question
8 months agoContributor-Level 10
To apply for IHM Hajipur scholarships, MSc students are required to complete the eligibility criteria and follow the application process. Check out the below pointers for better understanding:
Eligibility: Students belonging to SC, ST, and other eligible categories from Bihar are encouraged to apply for these scholarships.
Application Process: Interested students should apply through the Bihar Government's Welfare Department to avail of these financial assistance programs.
New Question
8 months agoNew Question
8 months agoContributor-Level 10
% of N = (1.4 × Normality of acid × volume of acid used) / (mass of organic compound)
42 = (1.4 × (1 × 2) × volume of acid used) / 0.8
Volume of acid used = (42 × 0.8) / (1.4 × 2) ml
New Question
8 months agoContributor-Level 10
The Delhi School of Communication offers students the popular programme of MBA in one specialisation. The total tuition fees for the course is INR 5.45 L. The various Delhi School of Communication MBA specialisation is mentioned below:
1. Master of Business Administration (MBA) (Part Time)
Students can opt for their preferred specialisation according to their particular career direction.
New Question
8 months agoContributor-Level 6
Some punctuations are categorized as end punctuations. The end punctuations are:
- Periods (.) – It is used to mark an end to the sentences.
- Question marks (? ) – It is used to end the sentence to indicate a query or direct question.
- Exclamation marks (!) – It is used to end the sentence to indicate emphasis or strong emotions.
New Question
8 months agoContributor-Level 10
Main product of electrolysis of conc. H? SO? is H? S? O? i.e. HO? SO-OSO? H
New Question
8 months agoContributor-Level 10
q = +150 J
w = -200 J
∴ ΔU = q + w = 150 - 200 = -50 J
So, magnitude of ΔU is 50 J.
New Question
8 months agoContributor-Level 9
(CH? )? N? HCl produces H? which reacts with NaHCO? and CO? gas evolved, because HCl is stronger acid than H? CO?
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts