Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Karnavati University offers BBA (Hons) through the Unitedworld Institute of Management (UIM). Admission to the programme is subject to the fulfilment of the eligibility criteria specified by the university. As per the university's official website, the eligibility requirement for admission to the BBA (Hons) programme of Karnavati University is as follows:
- Aspirants must have passed Class 12 or a three-year Diploma (equivalent to Class 12 from any recognised higher secondary boards as per the COBSE List)
- They must have scored a minimum aggregate of 55% in the qualifying exam
New Question
11 months agoContributor-Level 10
4.36 We know, The Arrhenius equation is given by k = Ae-Ea/RT Taking natural log on both sides,
Ln k = ln A- (Ea/RT)
Thus, log k = log A - (Ea/2.303RT). eqn 1
The given equation is log k = 14.34 – 1.25 * 104K/T. eqn 2
Comparing 2 equations, Ea/2.303R = 1.25 * 104K
Ea = 1.25 * 104K * 2.303 * 8.314
Ea = 239339.3 J mol-1 (approximately) Ea = 239.34 kJ mol-1
Also, when t1/2 = 256 minutes,
k = 0.693 / t1/2
= 0.693 / 256
= 2.707 * 10-3 min-1 k = 4.51 * 10-5s–1
Substitute k = 4.51 * 10-5s–1 in eqn 2,
log 4.51 * 10-5 s–1 = 14.34 – 1.25 * 104K/T
log (0.654-5) = 14.34– 1.25 * 104K/T = 1.25 * 104/ [ 14.34- log (0.654-5)] T = 668.9K or T =
New Question
11 months agoContributor-Level 10
4.35 The given equation is
k = (4.5 x 1011 s-1) e-28000K/T (i)
Comparing, Arrhenius equation
k = Ae -E/RT (ii)
We get, Ea / RT = 28000K / T
⇒Ea = R x 28000K
= 8.314 J K-1mol-1 × 28000 K
= 232792 J mol–1 or 232.792 kJ mol–1
New Question
11 months agoContributor-Level 10
4.34 t1/2 = 3.00 hours
We know, t1/2 = 0.693/k
? k = 0.693/3 k = 0.231 hrs-1
We know, time
Where, k- rate constant
[R]° -Initial concentration
[R]-Concentration at time 't'
Thus, substituting the values,
log ( [R]0/ [R]) = 0.8
log ( [R]/ [R]0) = -0.8
[R]/ [R]0 = 0.158
Hence, 0.158 fraction of sucrose remains.
New Question
11 months agoContributor-Level 10
4.33 Given,
k = 2.0 * 10–2s-1
time t = 100s
Concentration [A0] = 1.0 mol L-1
We know,
On substituting the values, Log (1/ [A]) = 2.303/2
Log [A] = -2.303/2 [A] = 0.135 mol L–1
New Question
11 months agoContributor-Level 10
4.32 Given,
k = 2.418 * 10-5 s-1
T = 546 K
Ea = 179.9 kJ mol-1 = 179.9 * 103J mol-1
The Arrhenius equation is given by k = Ae-Ea/RT Taking natural log on both sides,
Ln k = ln A- (Ea/RT) Substituting the values,
ln (2.418 * 10-5 ) = ln A-179.9/ (8.314 * 546)
ln A = 12.5917
A = 3.9 * 1012 s-1 (approximately)
New Question
11 months agoContributor-Level 10
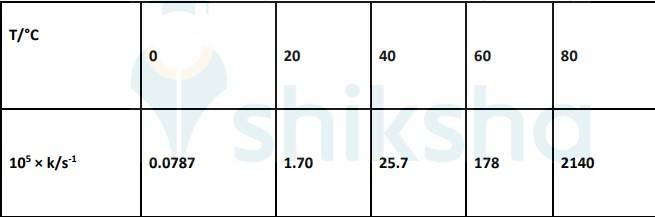
4.31 To convert the temperature in °C to °K we add 273 K.
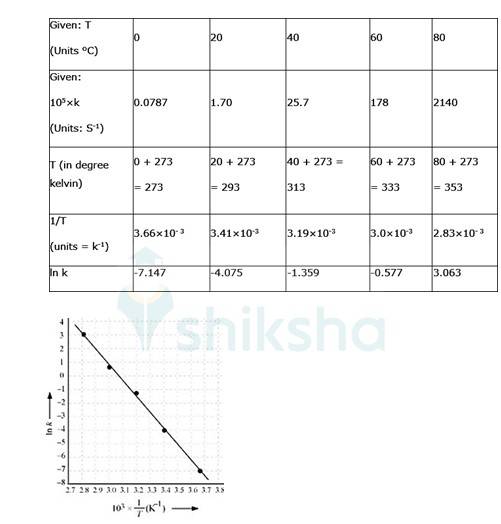
The graph is given as:
The Arrhenius equation is given by k = Ae-Ea/RT
Where, k- Rate constant
A- Constant
Ea-Activation Energy
R- Gas constant
T-Temperature
Taking natural log on both sides,
ln k = ln A- (Ea/RT). equation 1
By plotting a graph, ln K Vs 1/T, we get y-intercept as ln A and Slope is –Ea/R.
Slope = (y2-y1)/ (x2-x1)
By substituting the values, slope = -12.301
? –Ea/R = -12.301
But, R = 8.314 JK-1mol-1
? aE= 8.314 JK-1mol-1 * 12.301 K
? aE= 102.27 kJ mol-1
Substituting the values in equation 1 for data at T = 273K
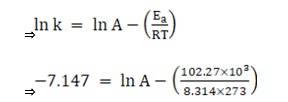
(? At T = 273K, ln k =-7.147)
On solving, we get ln A = 37.
New Question
11 months agoBeginner-Level 4
Actually percentile isn't calculated by marks it's based on your performance compared to other candidates.
Also, to know the exact percentile we need the official result data of your session but still we can calculate by checking previous year results and it would come around 70-80% and also it may vary based on other candidates results.
If you have further queries feel free to connect :)
New Question
11 months agoContributor-Level 10
4.30 When t = 0, the total partial pressure is P0 = 0.5 atm
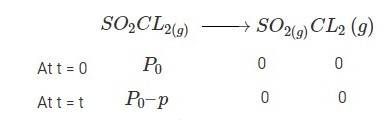
When time t = t, the total partial pressure is Pt = P0 + p
P0-p = Pt-2p, but by the above equation, we know p = Pt-P0
Hence, P0-p = Pt-2 (Pt-P0)
Thus, P0-p = 2P0 – Pt
We know that time
t= 2.303/K log R0 / R
Where, k- rate constant
[R]° -Initial concentration of reactant [R]-Concentration of reactant at time 't'
Here concentration can be replaced by the corresponding partial pressures.
Hence, the equation becomes,
t= 2.303/K log P0 / P0 - P
t= 2.303/K log P0 / 2P0 - Pt
? equation 1
At time t = 100 s, Pt = 0.6 atm and P0 = 0.5 atm,
Substituting in equation 1,
100 = 2.303/k log 0.5 /
New Question
11 months agoNew Question
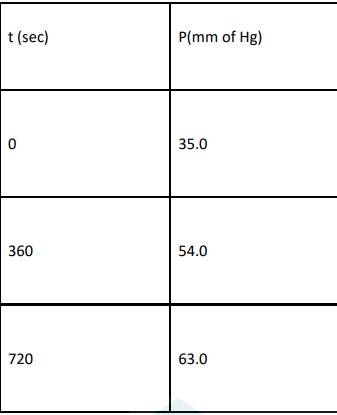
11 months agoContributor-Level 10
4.29 When t = 0, the total partial pressure is P0 = 35.0 mm of Hg
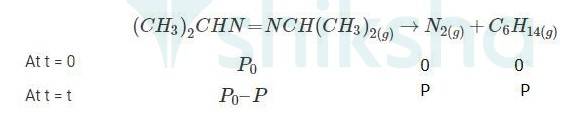
When time t = t, the total partial pressure is Pt = P0 + p
P0-p = Pt-2p, but by the above equation, we know p = Pt-P0 Hence, P0-p = Pt-2 ( Pt-P0)
Thus, P0-p = 2P0 – Pt
We know that time
t= 2.303/K log R0 / R
Where, k- rate constant
[R]° -Initial concentration of reactant
[R]-Concentration of reactant at time 't'
Here concentration can be replaced by the corresponding partial pressures.
Hence, the equation becomes,
t= 2.303/K log P0 / P0 - P
t= 2.303/K log P0 / 2P0 - Pt
? equation 1
At time t = 360 s, Pt = 54 mm of Hg and P0 = 30 mm of Hg, Substituting in equation 1,
360 = 2
New Question
11 months agoContributor-Level 10
Both Lakehead University as well as Thompson Rivers University offer globally recognised degrees. The differences are mentioned in the below table.
Parameters | Lakehead University | Thompson Rivers University |
|---|---|---|
World University Ranking | #1241 | #2294 |
Ranking for Programs | #325 | #60 |
Ranked in Canada | #36 | #55 |
Acceptance Rate | Around 82-90% | Around 82-84% |
Top Majors | Psychology, Civil Engineering, Computer Science, Mechanical Engineering, Computational Science | Arts, Law, Nursing, Science, Trades and Technology |
Duration of Courses | UG: 3 - 4 years PG: 1 - 2 years | UG: 4 - 5 years PG: 16 months - 2 years |
Annual Tuition Cost | UG: INR 19 L - 25 L PG: INR 4 L - 9.2 L | UG: INR 10 L - 15 L PG: INR 9.3L - 50.8L |
Application Fees | INR 8.3K | INR 8.5K |
Scholarships | UG: 3 PG: 2 | 6 |
New Question
11 months agoContributor-Level 10
There are many specialised courses offered by the top distance BA colleges in Bangalore. Some of the courses are mentioned below:
Top Specialisations | No. of Colleges |
|---|---|
English | 4 |
Languages | 4 |
Economics | 3 |
History | 3 |
Political Science | 3 |
New Question
11 months agoContributor-Level 10
Yes, there are many private distance BA colleges in Bangalore. Some of them are mentioned below along with their tuition fees:
Private Colleges | Tuition Fee |
|---|---|
INR 20,000 | |
INR 8,000 |
Disclaimer: This information is sourced from official website and may vary.
New Question
11 months agoContributor-Level 10
Yes, there are many affordable distance BA colleges in Bangalore. Some of them are mentioned below along with their tuition fees of less than INR 1 lakh:
Low-cost Colleges | Tuition Fee |
|---|---|
Jamia Millia Islamia - Centre for Distance and Open Learning Fees | INR 7,000 |
Aaruhi Institute Fees | INR 45,000 |
Wisdom School of Management, Bangalore Fees | INR 20,000 |
Geetanjali Institute Fees | INR 45,000 |
Bright International Educational Trust, Jayanagar Fees | INR 8,000 |
Disclaimer: This information is sourced from official website and may vary.
New Question
11 months agoContributor-Level 10
Students can check the basic eligibility criteria below for Distance BA course in Bangalore:
- Students must have passed Class 12 or equivalent in any stream from a recognised board of India.
- Many popular Distance BA colleges require at least 50% marks in Class 12 for accepting admission and may vary.
- There are no age limit criteria for Distance BA course.
New Question
11 months agoContributor-Level 10
As per popularity basis, listed below are the top distance BA colleges in Bangalore along with their tuition fees:
Top Colleges | Tuition Fee |
|---|---|
Directorate of Correspondence Courses and Distance Education, Bangalore University | INR 14,000 |
Center For Distance Education and Virtual Learning, Jain Deemed to be University | INR 27,000 – INR 38,000 |
Sri Sai Correspondence College | - |
Jamia Millia Islamia - Centre for Distance and Open Learning | INR 7,000 |
Mysore Correspondence College | - |
Disclaimer: This information is sourced from official website and may vary.
New Question
11 months agoContributor-Level 10
There are about 10+ distance BA colleges in Bangalore. Of these, 6 colleges are privately owned. Students can secure admission after passing Class 12 boards. Some of the top BA colleges in Bangalore for distance courses include Directorate of Correspondence Courses and Distance Education, Center For Distance Education and Virtual Learning, Sri Sai Correspondence College, Jamia Millia Islamia - Centre for Distance and Open Learning, and many others.
New Question
11 months agoContributor-Level 10
Yes, Karnavati University BSc (Hons) admissions 2025 are open. The university is currently accepting online applications for the academic year 2025-26. Interested candidates can visit the university's official website to apply for admission. Before applying, candidates must ensure they satisfy the course-specific eligibility criteria specified by the university. Further, candidates seeking admission based on their CUET UG scores must separately apply for the entrance exams through the official NTA website.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts