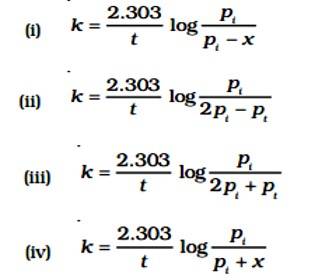Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Ans: Correct option A
Arrhenius equation Ae -
K = rate constant
A= frequency factor
Ea= Activation Energy
R= gas constant
T= temperature
ln k = lnA -
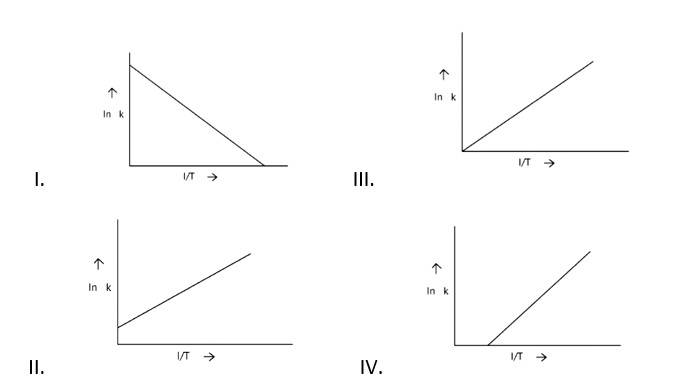
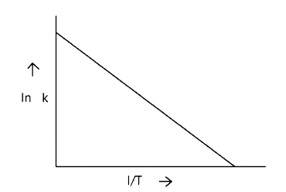
When the temperature rises, ln falls.
∴ ln kv/s , is a negative slope.
ln A is intercepted by k, and its magnitude decreases with time.
∴ Negative slope is obtained.
When compared to the other possibilities, T is increasing over time, which is incorrect.
ln k = + lnA
New Question
10 months agoGuide-Level 15
Yes, candidates have to fill out the application form for Faculty of Allied Health Sciences MRIIRS online. Aspirants can apply by paying an application fee of INR 1,200. Below are the steps to apply for admission:
Step 1: Visit the official website of MRIIRS.
Step 2: Click on 'Apply Now' on the Home page.
Step 3: Then Click on 'Apply Now' in front of the preferred MRIIRS institute/ faculty.
Step 4: Register with the basic details to generate login credentials.
Step 5: Login to fill out the application form and upload the documents.
Step 6: Pay the application fee to submit the form.
New Question
10 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Ans: Correct option B
Given:
A (g) → B (g) + C (g)
pi= Initial pressure
(Time) t = 0
A (g) → B (g) + C (g)
pi →0atm + 0atm
t, (pi - x)atm
pt = (pi - x)atm + x + x = pi + x
pA = (pi - x)
The value of x changes when it is substituted.
pA = pi - (pt - pi) = 2pi - pt
K = log
New Question
10 months agoContributor-Level 10
13.28 The equation for deuterium-tritium fusion is given as:
It is given that
Mass of ( = 2.014102 u
Mass of ( , 3.016049 u
Mass of ( = 4.002603 u
Mass of ( = 1.008665 u
Q-value of the given D-T reaction is:
Q =
=
= 0.018883
= 17.59 MeV
Radius of the deuterium and tritium, r = 2 m
Distance between the centers of the nucleus when they touch each other,
d = r +r = 4 m
Charge on the deuterium and tritium nucleus = e
Hence the repulsive potential energy between the two nuclei is given as:
V =
Where,
= permittivity of free space
It is given that = 9 N
Hence, V = 9 = 5.76 J =
New Question
10 months agoGuide-Level 15
Aspirants applying for admission at Faculty of Allied Health Sciences MRIIRS need to pay an application fee of INR 1,200. Candidates can pay the application fee via any of the online payment modes. Aspirants should keep the fee receipt with them for any future reference.
New Question
10 months agoContributor-Level 6
The SSC CHSL selection process comprises two tiers- Tier 1 and Tier 2. Candidates need to clear both the tiers to get shortlisted for appointment. Have a look at the details below:
- Tier I: Online objective-type exam
- Tier II: Online objective-type exam and skill test
The final allocation of candidates is made by indenting recruiting bodies.
New Question
10 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Ans: Correct option A
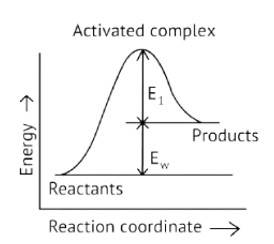
Activation Energy → The amount of energy required to overcome the obstacle and generate a product
The activation energy of a forward reaction can be seen in the [Eaf = E1 + E2] (This is an endothermic reaction.
Therefore, [Eaf > Eab]
The energy of the product is high, while the energy of the reactant is low.
The lower the energy, the more stable and positive the situation becomes.
Δ? = posistive
New Question
10 months agoContributor-Level 10
13.27 In the fission of , 10 particles decay from the parent nucleus. The nuclear reaction can be written as:
It is given that:
Mass of a = 238.05079 u
Mass of a =139.90543 u
Mass of a = 98.90594 u
Mass of a neutron , = 1.00865 u
Q value of the above equation,
Q =
Where
m’ = represents the corresponding atomic masses of the nuclei.
=
m’( =
m’( =
m’(
Substituting these values, we get
Q =
=
= u
=0.24807 u
= 231.077 MeV
New Question
10 months agoBeginner-Level 3
Tokyo is arguably the most expensive region to study in Japan, with average monthly cost of living (including food & other utilities) ranging between Yen 57,000-Yen 90,000 (INR 33.9 K-INR 53.6 K). Food, Transportation & other everyday utilities account for about INR 5 K, while the rest comes under rent & personal expenses. The table below gives a detailed breakdown of the living expenses students may incur over a month in Tokyo:
Product | Cost in Yen | Cost in INR |
|---|---|---|
Rice (5 kg) | 2,650 yen | INR 1.5 K |
| White Bread (1 kg) | 511 yen | INR 304 |
| Milk (1,000 ml) | 253 yen | INR 150 |
| Eggs (10 eggs) | 264 yen | INR 157 |
| Apples (1 kg) | 1,039 yen | INR 618 |
| Cabbage (1 kg) | 178 yen | INR 106 |
| Carbonated drink (1 liter) | 236 yen | INR 140 |
| Hamburger | 233 yen | INR 138 |
| Gasoline (1 liter) | 176 yen | INR 104 |
| Toilet paper (1,000 m) | 799 yen | INR 475 |
| Hair cut | 3,737 yen | INR 2.2 K |
| Taxi (4 km) | 1,693 yen | INR 1 K |
Total Cost | 9,110 Yen | INR 5.4 K |
Source: Portal Site of Official Statistics of Japan (Retail Price Survey of August 2024)
New Question
10 months agoGuide-Level 15
MRIIRS provides scholarships to students based on their MRNAT score. Students with a very high score can also get 100% fee waiver. Below are the details for the MRNAT scholarship available for Faculty of Allied Health Sciences MRIIRS students:
| MRNAT Score | 'Utkarsh' | 'Uttam' |
|---|---|---|
| 90% and above | 100% tuition fee waiver | 100% tuition fee waiver |
| 80% and above but below 90% | 50% tuition fee waiver | 25% tuition fee waiver |
| 70% and above but below 80% | 25% tuition fee waiver | 10% tuition fee waiver |
New Question
10 months agoContributor-Level 10
Generally, the National Institute of Pharmaceutical Education and Research does not accept direct admissions. Candidates must fulfill the requirements set forth by the university in order to be accepted into any of the programs offered. The procedure can be completed online. Candidates must pass a multi-step admissions procedure that consists of application, entrance, selection, and admission confirmation.
There is no provision of direct admission to NIPER, Ahmedabad.
New Question
10 months agoContributor-Level 10
13.26 For the emission of , the nuclear reaction is:
We know that:
Mass of , = 223.01850 u
Mass of , = 208.98107 u
Mass of , = 14.00324 u
Hence, the Q-value of the reaction is given as:
Q = ( - - )
= (223.01850 - 208.98107 - 14.00324) u
= 0.03419 u
= 0.03419 MeV = 31.848 MeV
Hence, the Q-value of the nuclear reaction is 31.848 MeV, since the value is positive, the reaction is energetically allowed.
For the emission of , the nuclear reaction is:
We know that:
Mass of , = 223.01850 u
Mass of , = 219.00948 u
Mass of , = 4.00260 u
Hence, the Q-value of the reaction is given as:
Q = ( - &n
New Question
10 months agoContributor-Level 10
The Staff Selection Commission (SSC) has announced a total of 3131 vacancies in the SSC CHSL 2025. These vacancies will be filled for the posts of Lower Division Clerk (LDC) and Data Entry Operator (DEO) in central government ministries and departments. Candidates who want to get recruited through the CHSL (10+2) exam need to apply online. The SSC CHSL applications were invited online.
New Question
10 months agoNew Question
10 months agoGuide-Level 15
Yes, candidates can get scholarship at Faculty of Allied Health Sciences MRIIRS based on their Class 12 percentage. MRIIRS provides Merit-based scholarships to students pursuing a course from the university. Below is the detailed criteria:
| Marks/ CGPA in Qualifying Examination | Tuition Fee Waiver |
|---|---|
| 98% and above | 100% |
| 95% to 97.99% | 50% |
| 90% to 94.99% | 25% |
| 85% to 89.99% | 10% |
New Question
10 months agoContributor-Level 10
13.24 If a neutron is removed from , the corresponding reaction can be written as:
+
The separation energies are
For : Separation energy = 8.363007 MeV
For : Separation energy = 13.059 MeV
It is given that
m( = 39.962591 u
m( = 40.962278 u
m( = 1.008665 u
The mass defect of the reaction is given as:
Δm = m ( m( m(
= 39.962591 + 1.008665 - 40.962278
= 8.978 u
=8.363007 MeV
For , the neutron removal reaction can be written as
+
It is given that
m( = 25.986895 u
m( = 26.981541 u
m( = 1.008665 u
The mass defect of the reaction is given as:
Δm = m( m(
New Question
10 months agoContributor-Level 10
13.15 The given nuclear reaction is
+ +
Atomic mass
m ( ) = 1.007825 u
m ( ) = 2.014102 u
m ( ) = 3.016049 u
The Q-value of the reaction can be written as:
Q =
=
= (-4.33 )
But 1 u = 931.5 MeV/
Q = -4.0334 MeV
The negative Q-value of the reaction shows that the reaction is endothermic.
The given nuclear reaction is
+ +
Atomic mass
m ( ) = 12.000000 u
m ( ) = 19.992439 u
m ( ) = 4.002603 u
The Q-value of this reaction is given as:
Q =
=
=4.958 u
=4.958
=4.6183 MeV
New Question
10 months agoGuide-Level 15
The total tuition fee for BSc/ BSc (Hons) at Faculty of Allied Health Sciences MRIIRS is INR 5.2 lakh. This information is sourced from official website/ sanctioning body and is subject to change. Aspirants who are shortlisted for admission have to pay the first installment of the fee to confirm their seat. The mode of payment is as specified by the university. Besides, aspirants also need to pay a registration fee of INR 1,200 in the start to apply for BSc/ BSc (Hons) programme.
New Question
10 months agoContributor-Level 10
13.13 The given values are
m ( = 11.011434 u and m ( ) = 11.009305 u
The given nuclear reaction:
Half life of nuclei, =20.3 min
The maximum energy possessed by the emitted positron = 0.960 MeV
The change in the Q-value (ΔQ) of the nuclear masses of the
ΔQ =
where
= Mass of an electron or positron = 0.000548 u
c = speed of the light
m’ = Respective nuclear masses
If atomic masses are used instead of nuclear masses, then we have to add 6 in the case of and 5 in the case of .
Hence the equation (1) reduces to
ΔQ =
=&nb
New Question
10 months agoGuide-Level 15
Candidates who have passed Class 12 with at least 50% aggregate in five subjects are eligible to apply for BSc/ BSc (Hons) programme at Faculty of Allied Health Sciences MRIIRS. Aspirants must hold the documents proving their eligibility for verification at the time of admission. Besides, the selection for BSc/ BSc (Hons) courses is based on merit or MRNAT scores of the candidates.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts