Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: The azo products have an extended conjugate system with both aromatic rings linked by the –N=N- bond. These compounds are frequently colored and used as dyes. Benzene diazonium chloride reacts with phenol to form p-hydroxy azobenzene by coupling the phenol molecule in its para position with the diazonium salt. This is referred to as a coupling reaction.
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: Amides are the byproducts of the acylation reaction. The reaction is carried out in the presence of a stronger base than the amine, such as pyridine, which removes the formed HCl and shifts the equilibrium to the right.
New Question
10 months agoContributor-Level 10
IIITDM Kurnool has emerged as one of the eminent Engineering Institutions in the state of Andhra Pradesh. The institute is granted the status of an Institution of National Importance and provides popular courses like BTech, MTech, MDes, PhD, among others. The infrastructure at IIITDM is top-notch and the placement landscape is impressive, with the 2024 IIITDM placements recording the highest package at INR 32.50 LPA.
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: As the electronegativity of oxygen is more than the electronegativity of a nitrogen atom, the O−H bond is more polar than the N−H bond, therefore MeOH is stronger acid than MeNH2 or MeNH2 is stronger base than MeOH.
New Question
10 months agoContributor-Level 10
Saint Joseph University in Philadelphia, USA, requires a strong academic record with a minimum GPA of 2.5 or greater on a scale of 4.0 for UG admissions.
Below are the general eligibility requirements for Saint Joseph's University for international students:
- Academic performance: A Minimum cumulative GPA of 2.5 or greater is required in post-secondary education. However, there are specific programme requirements mentioned for all sorts of programs.
- Transcripts: High school transcripts are needed to apply to SJU admissions in the USA.
- English proficiency: Minimum scores are needed for various English p
New Question
10 months agoContributor-Level 10
IIITDM Kurnool offers various scholarship initiatives that are designed to uplift students with weak educational backgrounds. Below mentioned are some of the prominent IIITDM Kurnool scholarship programs:
- Siemens Scholarship
- Micron Scholarship
- Reliance Foundation Scholarship
- Foundation for Excellence scholarship
- ONGC Scholarship
- Sitaram Jindal Scholarship
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: As −NH2 is a strong activating group, the aniline will readily undergo electrophilic substitution reaction, and it is difficult to cease reaction at the mono substitution stage.
Therefore, the activating group −NH2 is protected by an acetylation process.
The acetylated complex formed utilizes the lone pair of nitrogen and are less available for donation, this helps to carry out the nitration reaction easily.
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
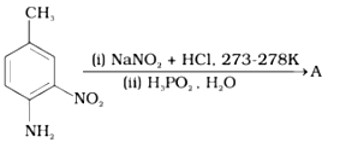
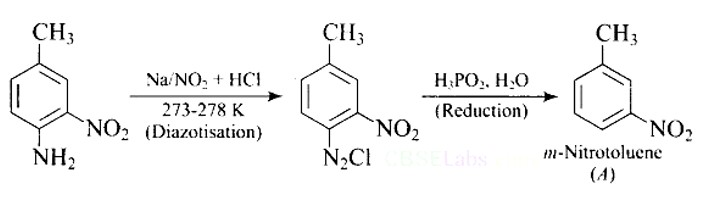
Ans: The reaction of aniline with nitrous acid at 273-278 K produces benzene diazonium chloride. The reaction of sodium nitrite with hydrochloric acid produces nitrous acid in the reaction mixture. Diazotisation is the process of converting primary aromatic amines into diazonium salts. Because of its instability, the diazonium salt is generally not stored or used immediately after preparation.
The crystalline solid benzene diazonium chloride is colorless.
It is easily soluble in water and stable at room temperature, but it reacts with water when warmed.
In the dry state, i
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
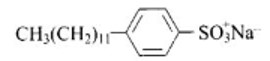
(a, d) Sodium salts of sulphonated long chain alcohol and sodium salts of sulphonated long chain hydrocarbons are anionic detergents e.g., Sodium laurylsulphate CH3 (CH2)10CH2OSO3– Na+ and sodium dodecylbenzene sulphonate.
New Question
10 months agoContributor-Level 10
The wide variety of courses that IIITDM offers are made available by the 4 distinct departments at the institute. Different BTech and M.Tech specializations are provided by the IIITDM faculty. Below are the departments at IIIT Kurnool:
Computer Science and Engineering | Electronics and Communication Engineering | Mechanical Engineering |
Department of Sciences | - | - |
New Question
10 months agoContributor-Level 10
As per reports, the highest stipend and average stipend offered were INR 3.95 Lacs per annum and INR 1.43 Lacs per annum, respectively for the PGP ABM batch. The complete details related to IIM Lucknow summer placements for PGP SM and PGP ABM are as follows:
Particulars | PGP SM Summer Placements (2023-25) | PGP/PGP ABM Summer Placements (2023-25) |
|---|---|---|
Total students | 44 | 576 |
Students participated | 43 | 576 |
Students placed | 43 | 576 |
Placement rate | 100% | 100% |
New recruiters | 15 | NA |
Companies participated | 22 | 210+ |
Average stipend | INR 1 Lacs per month | INR 1.30 Lacs per month |
the highest stipend | INR 4 Lacs per month | INR 3.5 Lacs per annum |
Median stipend | INR 1 Lacs per month | INR 1.31 Lacs per month |
Top 25% stipend | INR 1.73 Lacs per month | NA |
Top 50% stipend | INR 1.36 Lacs per month | NA |
*The stipend mentioned above is for the entire internship duration.
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: Hinsberg's reagent is also known as benzenesulphonyl chloride (C6H5SO2Cl). When it reacts with primary and secondary amines, it produces sulphonamides. Allowing secondary and tertiary amines to react with Hinsberg's reagent allows them to be distinguished (benzenesulphonyl chloride C6H5SO2Cl). Secondary amines react with Hinsberg's reagent to form an alkali-insoluble product. N, N-diethylamine, for example, reacts with Hinsberg's reagent to form N, N-diethylbenzenesulphonamide, which is insoluble in alkalis. Tertiary amines, on the other hand, are unaffected by Hin
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
A glycosidic linkage is a linkage that connects monosaccharides in polysaccharides. The covalent bond that joins two or more monosaccharides to produce a polysaccharide is known as this linkage.
New Question
10 months agoContributor-Level 10
Students seeking admissions to the various courses at IIITDM Kurnool can follow the below given steps:
- Visit the NTA official website (nta.ac.in).
- Fill out the NTA JEE application form.
- Log in and the completion of the application form.
- Upload the necessary scanned images and documents.
- Complete the application process by making the required payment start of the application fee.
New Question
10 months agoContributor-Level 10
Takshashila University MSc admissions are primarily merit-based. The admission committee selects students based on their performance in the qualifying examination. However, as per the admission, some students may be asked to appear in a written test or interview process. In such cases, the admissions committee will inform the students in a timely manner.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a, d) Tranquilizers are neurologically active drugs. Veronal and luminal are derivatives of barbituric acid used as tranquilizers.
New Question
10 months agoContributor-Level 10
Northeastern University is known for offering best degree programs at undergraduate and postgraduate levels for its international students. Northeastern University offers more than 300 programs across UG, PG, and doctorate levels. The university offers a wide array of specializations including Computer Science, Business, Health Sciences, and Engineering. Some top programs at Northeastern University that are ranked by Shiksha Popularity Rankings are listed below:
Top Program | Shiksha Popularity Ranking 2025 |
|---|---|
MS | #48 |
Civil Engineering | #32 |
Electrical Engineering | #40 |
LL.M. | #28 |
MBA/PGDM | #67 |
MIM | #62 |
BBA | #68 |
M.A. | #72 |
MSN | #76 |
New Question
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
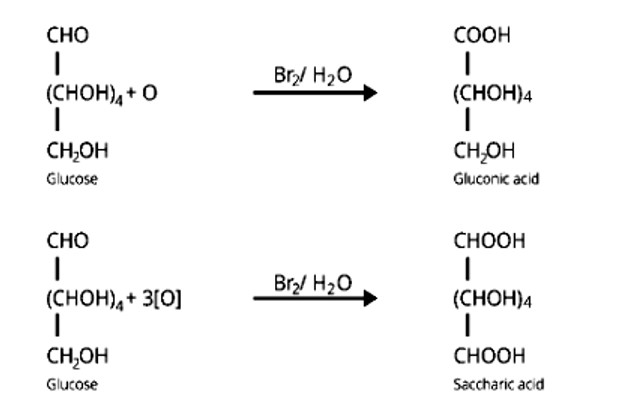
Ans:
New Question
10 months agoContributor-Level 10
IIITDM is located in a serene location in the town of Kurnool. The nearest airport to the institute campus is Rajiv Gandhi International Airport in Hyderabad, from where the total distance is around 210 kilometers. Students can hire a cab or opt for rail travel to reach the IIIT Kurnool campus. Kurnool Railway Station is located at about 25 kilometers from IIITDM and students can reach within a short time.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts