Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (ii)
Non-essential amino acids are amino acids that can be synthesized in the body and need not be taken from outside through diet. Glycine is an example of non-essential amino acids and is available in plenty of foods.
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (ii)
When DNA (or RNA) is completely hydrolyzed, it produces a 5 carbon based sugar called pentose sugar, phosphoric acid, and nitrogen-containing heterocyclic molecules (called bases). The sugar moiety of the nucleic acid, DNA is β−D−2− deoxyribose. Therefore, both assertion and reason are wrong statements.
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (v)
All other naturally occurring alpha-amino acids, with the exception of glycine, are optically active due to the asymmetry of the α - carbon atom. Both of these ' D ' and ' L ' configurations are available. The majority of amino acids found in nature have a L− configuration. The group on the left side is used to designate L− amino acids.
naturally occurring amino acids have a configuration. The −NH2 group on the left side is used to indicate amino acids.
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (iv)
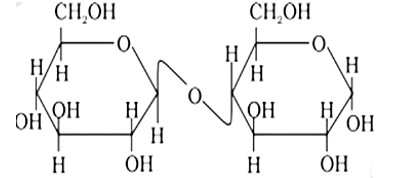
Maltose is made up of two? D? glucose units, one of which, C?1, is coupled to the C?4 of the other one. As a result, maltose contains a? glycosidic linkage. As a result, the assertion is a false statement, but the reason is true.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
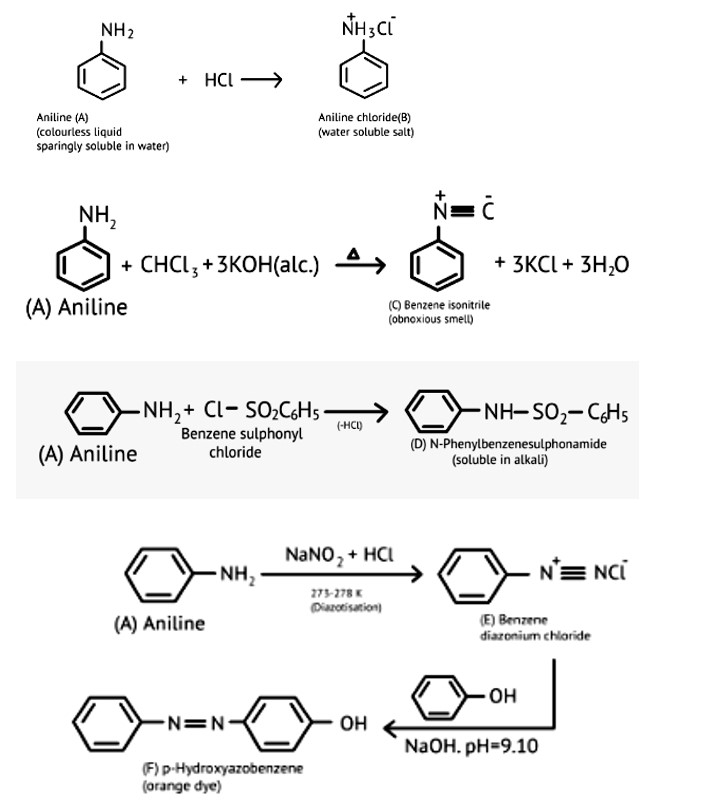
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
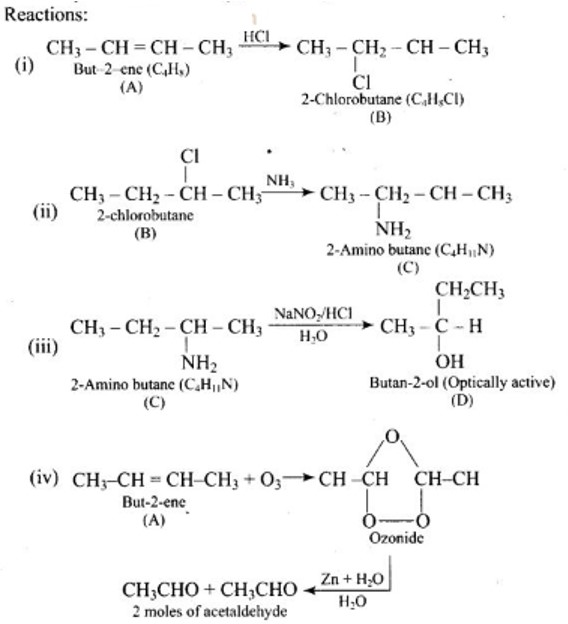
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (i)
Vitamins that are fat soluble are soluble in fats and oils but insoluble in water. They can be stored in the liver as well as adipose (fat-storing) tissues and are not excreted in urine . As a result, both assertion and reason are true assertions, and reason clarifies the assertion.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (d)
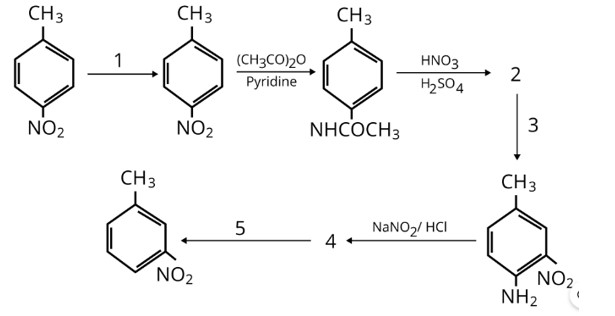
Acetanilide is less basic than aniline because electron density of nitrogen is lowered by acetyl group.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (a)
Aromatic primary amines cannot be prepared using Gabriel phthalimide synthesis as aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide. Both the statement's assertion and reason are incorrect, hence the correct answer is a.
New Question
10 months agoContributor-Level 10
This is an Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (iii)
Because D (+) - glucose causes plane polarised light to rotate to the right, it is
dextrorotatory. D denotes the relative configuration of glucose in regard to
glyceraldehyde in this situation. As a result, assertion is the proper statement, while
reason is the incorrect statement.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (d)
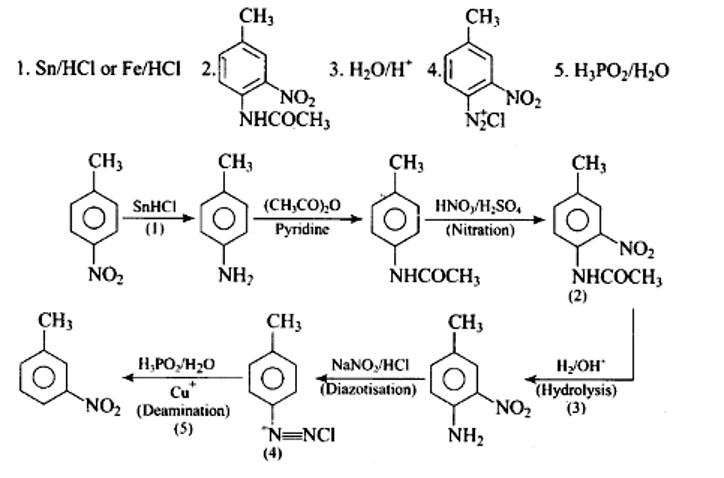
Fe+2HCl→FeCl2+2 [H] FeCl2+H2O (g)→FeO+2HCl
The nascent hydrogen formed act as reducing agent for the reduction of nitro compounds.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (b)
N, N-die ethyl benzene sulphonamide, there is no acidic hydrogen present on the N-atom which can make it soluble. Therefore, it is insoluble in alkali.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (d)
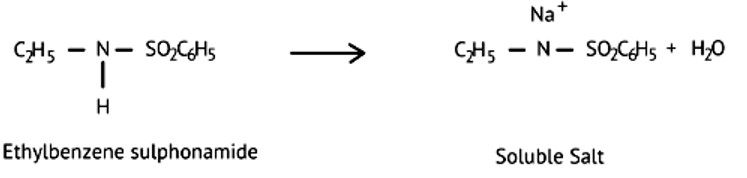
Ethylbenzene sulphonamide is soluble in alkali because it has acidic hydrogen.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (a)
In Hoffmann bromamide degradation reaction as in this reaction primary amide group is treated with halogen first bromine then the halogen substituted amide product is coverts to primary amine with the release of carbon dioxide gas. Both the statements assertion and reason are incorrect therefore the correct option is a.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (c)
In the acryl derivative, the delocalisation of electrons of the nitrogen atom occur over the carbonyl group, this decreases the electron density on the nitrogen atom that it no more perform as nucleophile and don't react with next acylating alkaline molecule.
Therefore, Assertion statement is correct but reason is incorrect.
New Question
10 months ago
Contributor-Level 10
As per the latest report, the average package offered during Chouksey Engineering College placements in 2025 stood at INR 5 LPA. The same is tabulated below:
Particulars | Placement Statistics (2025) |
|---|---|
Average package | INR 5 LPA |
New Question
10 months agoContributor-Level 10
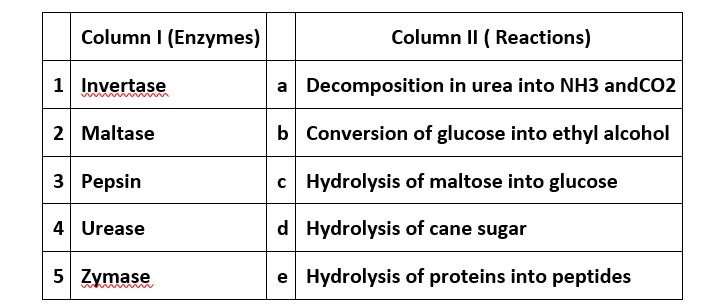
This is a Matching Answer Type Questions as classified in NCERT Exemplar
(i) → (d) (ii) → (c) (iii) → (e) (iv) → (a) (v) → (b)
New Question
10 months agoContributor-Level 10
BSc at Gangadhar Meher University is offered as a on campus programme. Students need to pay the Gangadhar Meher University course fee as against the opted curriculum to the institution. The Gangadhar Meher University BSc fees is in the range of INR 10,680 - 90,000. This fee is taken from the official website/sanctioning body. It is still subject to changes and hence, is indicative.
New Question
10 months agoContributor-Level 10
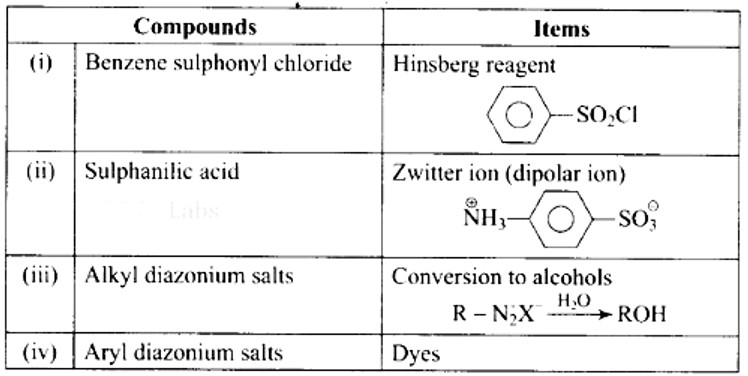
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)- (b), (ii)- (a), (iii)- (d), (iv)- (c)
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts