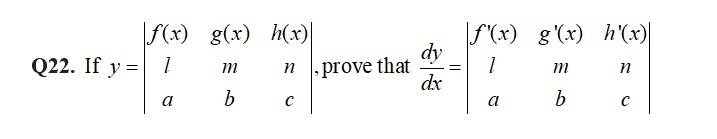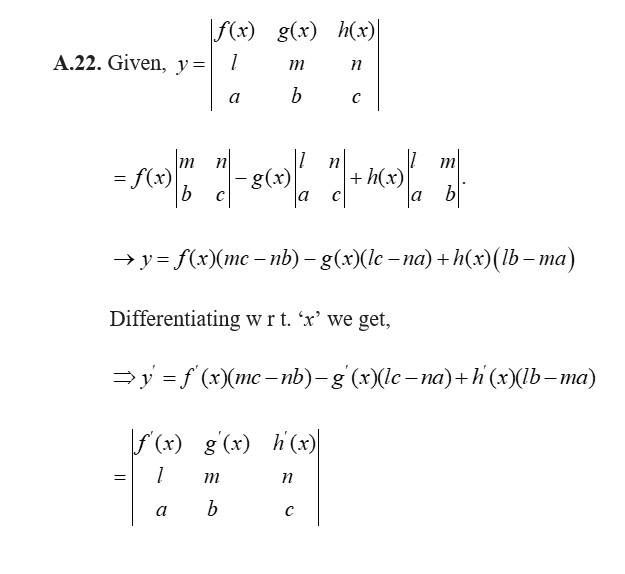Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The Pearl Academy Mumbai offers various specialisations for its BBA programme for a course duration of three years. The selection and eligibility for all specialisations are the same. Candidates can refer to the following list of specialisations offered at Pearl Academy Mumbai:
- Global Luxury Brand Management
- Fashion & Lifestyle Business Management
- BBA (with a choice of 5 specialisations, such as Advertising and Brand Management, Digital Communication Management, Data Science and Business Analytics, Media and Entertainment Management, and Human Resource Management)
New Question
10 months agoContributor-Level 10
There is no age limit as such for pursuing BA Public Administration. Complete your higher secondary education and secure a minimum of 50% aggregate in any stream and you'll easily secure your seats in your desired college. Admission is both merit-based and entrance-based.
New Question
10 months agoContributor-Level 10
The given D.E is
Which is of form
So,
Thus the general solution is of the form,

New Question
10 months agoContributor-Level 10
12. When tetravalent germanium is doped with trivalent gallium then some of the positions of the lattice of germanium gets occupied by the gallium. Since the gallium atom has only three valence electrons, the fourth valency of the nearby germanium atom does not get satisfied and hence this place remains vacant. This place is deficient of electrons and is therefore called an electron hole or electron vacancy.
Now, the electron from the neighbouring atom comes and fills the gap and leads to the formation of a hole in its original position. Under the influence of electric fields, the electrons move towards the positively charged plat
New Question
10 months agoContributor-Level 10
The given D.E is

Which is of form
So,
Thus the solution is of the form.
New Question
10 months agoContributor-Level 10
The given D.E. is
Which is of form
So ,
Thus the solution is of the form,
New Question
10 months agoContributor-Level 10
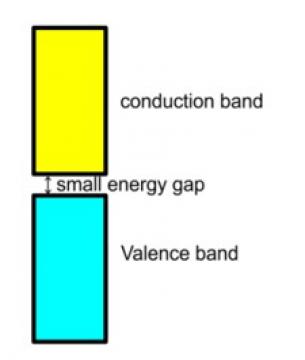
11. In semiconductors the gap between conduction band and valence band is small and hence some of the electrons from the valence band can easily jump to the conduction band and shows some conductivity but with rise in the temperature more of the electrons gets jump to the conduction band and thus, their electrical conductivity increases with rise in the temperature.
New Question
10 months agoContributor-Level 10
Marwari College in Ranchi has a well-structured placement cell that frequently invites various industrial and corporate organizations from across India for campus recruitment drives aimed at final year UG/PG students. The placement cell operates under the leadership of Principal and Convenor, along with a team of faculty members and staff from different departments. They work together with the visiting teams around the clock to ensure the success and support of the campus placement drives.
New Question
10 months agoContributor-Level 10
Motherhood University has a total of 60 seats for the BPharm programme. Candidates are admitted as per the sanctioned intake and selection rounds. The university reserves the right to revise the seat count in an academic year. It must be noted that the mentioned seat intake is as per the official website/ sanctioning body. It is however still subject to changes and hence, is indicative.
New Question
10 months agoContributor-Level 10
The given D.E. is
Which is of form
So,
Thus, the general solution is of the form
New Question
10 months agoContributor-Level 10
SRM University has a good record for MBA placements. As per the available information, the average package bagged by the students during MBA placements in 2023 was INR 7.75 LPA. Other than this, earlier in 2022, the highest package and average packages offered to MBA students in 2022 stood at INR 31 LPA and INR 7.75 LPA, respectively.
New Question
10 months agoContributor-Level 10
10. The ZnO crystal becomes yellow oh heating because of the metal excess defect which is caused due to the presence of extra cations at the interstitial sites and on heating this white crystal it loses oxygen and turns yellow. The reaction involved is given as-
ZnO Zn2+ + O2 + 2e-
Here the excess of Zn2+ ions move to the interstitial sites and electrons to neighbouring interstitial sites.
New Question
10 months agoContributor-Level 10
Marwari College, Ranchi provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies and it is totally worth it. Check out the table below to know the key highlights of the Marwari College, Ranchi placements:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 9 LPA |
Top Recruiters | TCS, Wipro, HDFC Bank, etc. |
Students Placed | 450 |
Note- The institute has not released the 2024 placement report, once released it will be updated here.
New Question
10 months agoContributor-Level 10
The given D.E. is
which is of form
So,
Thus, the general solution is of the form.
New Question
10 months agoContributor-Level 10
The course-wise placements for Marwari College has not been disclosed as of yet. Meanwhile, check out the table below to know the key highlights of Marwari College, Ranchi placement in 2023:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 9 LPA |
Top Recruiters | TCS, Wipro, HDFC Bank, etc. |
Students Placed | 450 |
Note- The institute has not released the 2024 placement report, once released it will be updated here.
New Question
10 months agoContributor-Level 10
So, x = 1, x= 2 divides the real line into three disjoint intervals and
For
For
For
Hence, these polynomial fun are all continous and desirable. for all real values of x or, except x = 1 and x = 2.
ie,
For differentiavity at x = 1,
LHD =
= -2
RHD =
as L.HD ≠ R.HD
f is not differentiable at x =1.
For continuity at x = 1.
L.HL=
RHL = \ LHL = RHS
f is continuous at x = 1
For continuity & differentiability at x = 2
? LHL = RHL
f is continuous at x = 2
= 2
? LHD ≠ RHD
f is not differentiable at x = 2.
New Question
10 months agoContributor-Level 10
Marwari College, Ranchi provides good placements to its graduating students. Check out the table below to know the key highlights of the Marwari College, Ranchi placement in 2023:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 9 LPA |
Top Recruiters | TCS, Wipro, HDFC Bank, etc. |
Students Placed | 450 |
New Question
10 months agoContributor-Level 10
The given D.E.is
Which is of form
So,
Thus, the general solution is of the form.
Let,
New Question
10 months agoContributor-Level 10
9. In FeO crystal, some of the Fe2+ ions are replaced by Fe3+ ions i.e., 3Fe2+ ions are replaced by 2Fe3+ ions to make up for the loss of positive charge. As a result of which it leads to lesser amount of metal as compared to the stoichiometric proportion.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts