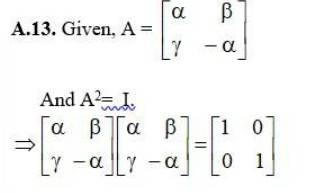Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Given, A2= A.
(E) we need to calculate,
(I + A)3- 7A = I3 + A3 + 3IA (I + A) - 7A { (x + y)3 = x3 + y3 + 3xy (a + y)}

New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a multiple choice type question as classified in NCERT Exemplar
Correct option (i) and (ii)
On moving down the group, as the size increases the hydration enthalpy decreases. The hydration enthalpy of beryllium and magnesium ions are high due to the small size. Thus, sulfates are readily soluble in water.
New Question
10 months agoContributor-Level 10
Given, A is both symmetric and skew-symmetric.
(E) Then, A' = A ____ (1) and A' = -A ____ (2)
So using (2), A' = -A.
A = -A {eqn (I)}
A + A = 0
2A = 0
A = 0.
A is a zero matrix
So, option B is correct.
New Question
10 months agoContributor-Level 10
The candidates who want to study a BDes course can compare different parameters in order to decide which university is better than Pearl Academy Mumbai. The number of specialisations offered, student ratings, amount of fees, placement reports, return on investment (ROI), faculty qualifications, and other similar parameters are some of the most important factors to consider. After careful consideration of these parameters, the candidates can choose their preferred course of study.
New Question
10 months agoContributor-Level 10
This is a multiple choice type question as classified in NCERT Exemplar
Correct option (i) and (iii)
Na2CO3 is used in paper paints and textile industry. It is used to manufacture soap powders.
NaOH is used in textile industry for mercerising cotton fabrics.
New Question
10 months agoContributor-Level 10
Yes, at Maya Somaiya School of Music and Performing Arts, students who are officially pursuing a MPA course are offered scholarships. The institute has various types of scholarships to cater to a wide range of student needs. The below list contains some of the scholarships offered by the institute:
- EWS (Economically Weaker Section) Scholarship
- For Students with Disabilities (PWD)
- Sports Merit Scholarship
New Question
10 months agoContributor-Level 10
This is a multiple choice type question as classified in NCERT Exemplar
Correct option (ii) and (iv)
Alkali metals have larger atomic size and low density. These metals lose electrons due to less effective nuclear charge. They have a high value of electrode potential.
Lithium has the highest negative reduction potential value. Due to the small size of lithium it has a small atomic size and the highest ionization enthalpy.
New Question
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
(i) Grp-13 The atomic size of the boron family follows the irregular trend. Generally, down the group the size increases but Gallium has a smaller atomic radius than Aluminium due to the poor shielding effect of 3d-orbitals.
Order: B
Grp-14 The size of the carbon family is smaller than the modern family and as we move down the group the atomic size increases regularly. The increase in covalent radius from carbon to silicon is prominent while from Silicon to lead a small increase in covalent radius is observed; this is due to the presence of completely filled D and f-o
New Question
10 months agoContributor-Level 10
After completing an MBA course from SRM School of Management, candidates can pursue various job opportunities in the fields of Management, Marketing, Sales, etc. Moreover, the course also prepares the students with the knowledge, skills, and strategic perspectives essential to business leadership and a managerial career in the competitive world. Some of the job roles that candidates can pursue after completing the course are as follows:
- International Marketing Manager
- Global Business Consultant
- Foreign Trade Analyst
- Human Resource Manager
- Business Development Manager, etc.
New Question
10 months agoContributor-Level 10
Micheal Burns, an American entertainment executive & the vice chairman of Lionsgate Productions, is a notable alumni of the ASU Engineering School.
Lionsgate is a famous entertainment organizations known for producing & distributing a number of movies & TV Shows. Among some of its popular franchises are: The Hunger Games, John Wick, Twilight & Saw.
Some other popular movies produced by it includes: Rambo, The Expendables, Hacksaw Ridge & the Blair Witch project.
Micheal pursued a Bachelor of Science degree from the institute in 1980 before going on to attend UCLA in 1992 for his MBA studies.
New Question
10 months agoContributor-Level 10
This is a multiple choice type question as classified in NCERT Exemplar
Correct option (iv)
Chlorides of alkaline earth metals are hydrated salts. They can be used as dehydrating agents due to their hygroscopic nature to absorb moisture from air. Extent of hydration decreases from Mg to Ba.
Therefore, dehydration of hydrates of halides of calcium, barium and strontium i.e., CaCl2.6H2O, BaCl2.2H2O, SrCl2.2H2O can be achieved by heating.
New Question
10 months agoContributor-Level 10
Yes candidates can join UHS Bagalkot BTech if they do not want to give the KCET entrance exam. Candidates can apply through the ICAR AIEEA entrance exam to secure admission into the programme. Once the results of either of the exams are announced, candidates can apply for admission into the programme through the university website.
New Question
10 months agoContributor-Level 10
Yes, BVIMSR admission is open for the current year. The institute offers MMS course at the PG level. Candidates looking for course admission must meet the eligibility criteria set by the college, Aspirants must pass graduation with a 45%-50% aggregate. Student appearing for final year examinations can also apply, but their admission will be subject to obtaining a minimum of 50% aggregate (45% for SC/ST) at qualifying examination. Diploma holders are not eligible.
New Question
10 months agoContributor-Level 10
Yes, fees play an important role when comparing the MPA course offered by Maya Somaiya School of Music and Performing Arts with other similar institutes/colleges. Drawing a comparison based on fees helps students calculate the affordability of a program. The lower the fees, the greater the affordability. As per official sources, the total tuition fees to pursue the MPA course at the institute is INR 80,000.
Note: The above-mentioned fee is as per the official sources. However, it is indicative and subject to change.
New Question
10 months agoContributor-Level 10
We have, AB = BA. (given)
(E) P (n):AB’ = B’A.
P (i):AB1 = B1A. Þ AB = BA
so, the result is true for n = 1.
Let the result be true for n = k.
P (k):ABk = BkA
Then,
P (k + 1) : ABk + 1 = A. Bk. B = BkA.B = Bk.BA
= Bk + 1.A .
So, ABk + 1 = Bk + 1A.
The result also holds for n = k + 1.
Hence, AB^n = B^n A^n holds for all natural number ‘n’.
New Question
10 months agoContributor-Level 10
This is a multiple choice type question as classified in NCERT Exemplar
Correct Option (iii)
The chemical formula of A is Ca (OH)2 Sodium carbonate is prepared by Solvay's process. In this process, ammonia is recovered when ammonium chloride reacts with calcium hydroxide. On reacting NH4Cl with Ca (OH)2, calcium chloride is obtained as a by-product. The reaction is given below.
2NH4Cl+Ca (OH)2→2NH3+CaCl2+H2O
When CO2 is bubbled through an aqueous solution of Ca (OH)2 the solution turns milky. It is used in whitewashing due to its disinfectant nature.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts