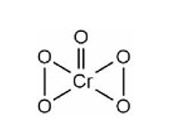
The addition of dilute NaOH to Cr3+ salt solution will give:
The addition of dilute NaOH to Cr3+ salt solution will give:
Option 1 - <p>Precipitate of <!-- [if gte mso 9]><xml>
<o:OLEObject Type="Embed" ProgID="Equation.DSMT4" ShapeID="_x0000_i1025"
DrawAspect="Content" ObjectID="_1815919415">
</o:OLEObject>
</xml><![endif]--> <span class="mathml" contenteditable="false"> <math> <mrow> <msup> <mrow> <mrow> <mo>[</mo> <mrow> <mi>C</mi> <mi>r</mi> <msub> <mrow> <mrow> <mo>(</mo> <mrow> <mi>O</mi> <mi>H</mi> </mrow> <mo>)</mo> </mrow> </mrow> <mrow> <mn>6</mn> </mrow> </msub> </mrow> <mo>]</mo> </mrow> </mrow> <mrow> <mn>3</mn> <mo>−</mo> </mrow> </msup> </mrow> </math> </span></p>
Option 2 - <p>Precipitate of <!-- [if gte mso 9]><xml>
<o:OLEObject Type="Embed" ProgID="Equation.DSMT4" ShapeID="_x0000_i1025"
DrawAspect="Content" ObjectID="_1815919431">
</o:OLEObject>
</xml><![endif]--> <span class="mathml" contenteditable="false"> <math> <mrow> <mi>C</mi> <msub> <mrow> <mi>r</mi> </mrow> <mrow> <mn>2</mn> </mrow> </msub> <msub> <mrow> <mi>O</mi> </mrow> <mrow> <mn>3</mn> </mrow> </msub> <msub> <mrow> <mrow> <mo>(</mo> <mrow> <msub> <mrow> <mi>H</mi> </mrow> <mrow> <mn>2</mn> </mrow> </msub> <mi>O</mi> </mrow> <mo>)</mo> </mrow> </mrow> <mrow> <mi>n</mi> </mrow> </msub> </mrow> </math> </span></p>
Option 3 - <p>Precipitate of <!-- [if gte mso 9]><xml>
<o:OLEObject Type="Embed" ProgID="Equation.DSMT4" ShapeID="_x0000_i1025"
DrawAspect="Content" ObjectID="_1815919449">
</o:OLEObject>
</xml><![endif]--> <sup><span class="mathml" contenteditable="false"> <math> <mrow> <mi>C</mi> <mi>r</mi> <msub> <mrow> <mrow> <mo>(</mo> <mrow> <mi>O</mi> <mi>H</mi> </mrow> <mo>)</mo> </mrow> </mrow> <mrow> <mn>3</mn> </mrow> </msub> </mrow> </math> </span></sup></p>
Option 4 - <p>A solution of <!-- [if gte mso 9]><xml>
<o:OLEObject Type="Embed" ProgID="Equation.DSMT4" ShapeID="_x0000_i1025"
DrawAspect="Content" ObjectID="_1815919455">
</o:OLEObject>
</xml><![endif]--> <span class="mathml" contenteditable="false"> <math> <mrow> <msup> <mrow> <mrow> <mo>[</mo> <mrow> <mi>C</mi> <mi>r</mi> <msub> <mrow> <mrow> <mo>(</mo> <mrow> <mi>O</mi> <mi>H</mi> </mrow> <mo>)</mo> </mrow> </mrow> <mrow> <mn>4</mn> </mrow> </msub> </mrow> <mo>]</mo> </mrow> </mrow> <mrow> <mo>−</mo> </mrow> </msup> </mrow> </math> </span></p>
4 Views|Posted 9 months ago
Asked by Shiksha User
1 Answer
A
Answered by
9 months ago
Correct Option - 2
Detailed Solution:
Kindly go through the solution
Similar Questions for you
K2Cr2O7 + H2O2 + H2SO4->
Potassium permanganate in alkaline medium oxidise lodide to lodate.
Compound A is
KMnO4 decomposes upon heating at 513 K and forms K2MnO4 and MnO2.
2KMnO4
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering