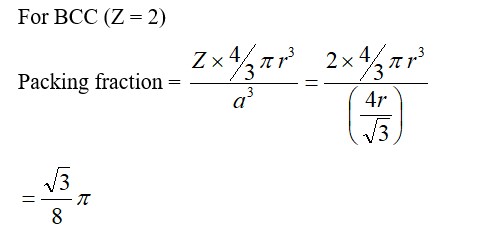1.33 Explain (i) The basis of similarities and differences between metallic and ionic crystals. (ii) Ionic solids are hard and brittle.
1.33 Explain (i) The basis of similarities and differences between metallic and ionic crystals. (ii) Ionic solids are hard and brittle.
(i) Both metallic and ionic crystals have strong forces of attraction between their atoms or ions. In metallic crystals there is a strong metallic bond present between electrons and positively charged ions. In ionic crystals there are strong ionic bonds between anions and cations. Both of them cond
Similar Questions for you
ΔG° = –RT * 2.303 log K
–nFE° = +RT * 2.303 log K
2 * 96500 * 0.295 = 8.314 * 298 * 2.303 log10 K
10 = log10 K = 1010
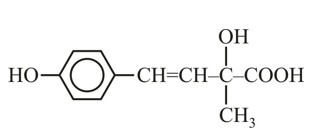
It has chiral centre and differently di substituted double bonded carbon atoms.
For FCC lattice
Packing efficiency = 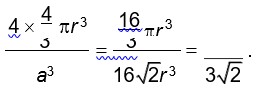
CsCl has BCC structure in which Cl– is present at corners of cube and Cs+ at body centre
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering