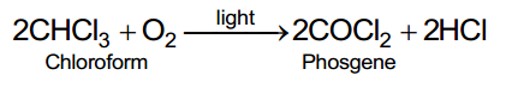
10.13 Which one of the following has the highest dipole moment?
(i) CH2Cl 2 (ii) CHCl 3 (iii) CCl4
10.13 Which one of the following has the highest dipole moment?
(i) CH2Cl 2 (ii) CHCl 3 (iii) CCl4
202 Views|Posted a year ago
Asked by Shiksha User
1 Answer
V
Answered by
10 months ago
Dipole moment of a molecule depends on the electronegativity difference between atoms bonded covalently and geometry of the molecule (how far the atoms are from each other). Dipole moment is important to understand the polarity of a molecule.
The three dimensional structures of the three compounds al
Similar Questions for you
Photodiode in reverse bias mode is used as intensity measuring device.
Tertiary haloalkane does not undergo SN2 reaction
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering