10.14 A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
10.14 A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
As per the molecular formula , one thing can be confirmed; the hydrogen is either cycloalkane or it is an alkene. However, alkenes readily react with chlorine in even dark because of the double bond. Therefore, it is possible that the hydrocarbon is cycloalkane.
Now, let us consider the reactivity
Similar Questions for you
Photodiode in reverse bias mode is used as intensity measuring device.
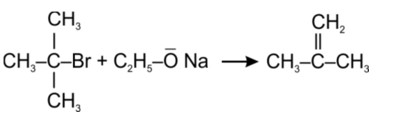
Tertiary haloalkane does not undergo SN2 reaction
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering