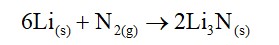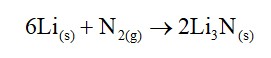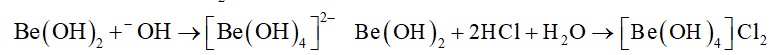10.26. Comment on each of the following observations:
(a) The mobilities of the alkali metal ions in aqueous solution are Li+ < Na+ + < Rb+ < Cs+
(b) Lithium is the only alkali metal to form a nitride directly.
(c) E? for M2+ (aq) + 2e– → M(s) (where M = Ca, Sr, or Ba) is nearly constant.
10.26. Comment on each of the following observations:
(a) The mobilities of the alkali metal ions in aqueous solution are Li+ < Na+
(b) Lithium is the only alkali metal to form a nitride directly.
(c) E? for M2+ (aq) + 2e– → M(s) (where M = Ca, Sr, or Ba) is nearly constant.
(a) Smaller the size of the ion, more highly it is hydrated and hence greater is the mass of the hydrated ion and thus the ionic mobility become lesser. The extent of hydration decreases in the order.
Li+ < Na+
Thus the mobility of Cs+ will be the highest
(b) Due to its smaller size lithium can form n
Similar Questions for you
Li+ has the highest hydration enthalpy.
Hence it is most hydrated
Therefore, Correct order of hydrated radii is Cs+ < Rb+ < K+ < Na+ < Li+
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering