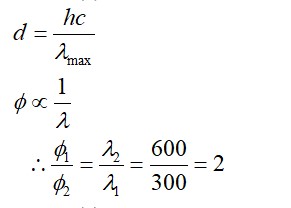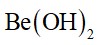10.29. How would you explain the following observations?
(i) BeO is almost insoluble but BeSO4 is soluble in water,
(ii) BaO is soluble but BaSO4is insoluble in water,
(iii) LiI is more soluble than KI in ethanol.
10.29. How would you explain the following observations?
(i) BeO is almost insoluble but BeSO4 is soluble in water,
(ii) BaO is soluble but BaSO4is insoluble in water,
(iii) LiI is more soluble than KI in ethanol.
(i) Lattice energy of BeO is comparatively higher than the hydration energy. Therefore, it is almost insoluble in water. Whereas, BeSO4 is ionic in nature and its hydration energy dominates the lattice energy.
(ii) Both BaO and BaSO4 are ionic compounds but the hydration energy of BaO is higher tha
Similar Questions for you
Li+ has the highest hydration enthalpy.
Hence it is most hydrated
Therefore, Correct order of hydrated radii is Cs+ < Rb+ < K+ < Na+ < Li+
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering