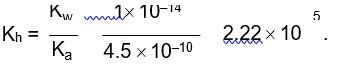200mL of 0.01 M HCl is mixed with 400mL of 0.01 M H2SO4. The pH of the mixture is___________.
Given: log2 = 0.30, log 3 = 0.48, log5 = 0.70, log7 = 0.84, log11 = 1.04
200mL of 0.01 M HCl is mixed with 400mL of 0.01 M H2SO4. The pH of the mixture is___________.
Given: log2 = 0.30, log 3 = 0.48, log5 = 0.70, log7 = 0.84, log11 = 1.04
Option 1 - <p>1.14 </p>
Option 2 - <p>1.78 </p>
Option 3 - <p>2.34 </p>
Option 4 - <p>3.02</p>
24 Views|Posted 10 months ago
Asked by Shiksha User
1 Answer
P
Answered by
10 months ago
Correct Option - 2
Detailed Solution:
Total concentration of H+ after the mixing of HCl and H2SO4;
Similar Questions for you
0.01 M NaOH,
M = 1 * 10-2
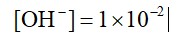
pOH = 2
pH = 2
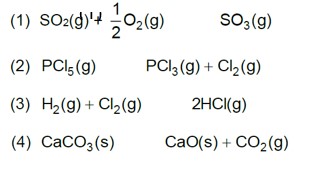
Kp = Kc (RT)Dng
36 * 10–2 = Kc (0.0821 * 300)–1
Kc = 0.36 * 0.0821 * 300 = 8.86 » 9
A(g) ->B(g) + (g)
Initial moles n 0 &nbs
On increasing pressure, equilibrium moves in that direction where number of gaseous moles decreases.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
7L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering