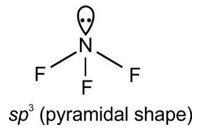
4.45. Assertion: In PCl5, the axial bonds are slightly longer and hence slightly weaker than the equatorial bonds.
Reason: The axial bond pairs suffer more repulsive interaction from the equatorial bond pairs.
4.45. Assertion: In PCl5, the axial bonds are slightly longer and hence slightly weaker than the equatorial bonds.
Reason: The axial bond pairs suffer more repulsive interaction from the equatorial bond pairs.
4.45. (a) It should be noted that all the bond angles in trigonal bipyramidal geometry are not equivalent. In PCl5 the five sp3d orbitals of phosphorus overlap with the singly occupied
p orbitals of chlorine atoms to form five P–Cl sigma bonds. Three P–Cl bond lie in one plane and make an angle of 1
Similar Questions for you
He2 has zero bond order hence it does not exist.
The three fundamental laws of chemistry are - Law of Definite Proportions, Law of Conservation of Mass, and Law of Multiple Proportions.
The three types of chemical bonds are - ionic, metallic and covalent bonds. When the electrons transfer between the atoms, they form the Ionic bonds by producing charged ions that are attracted to each other. When atoms share electrons, covalent bonds are created. When metal atoms share a sea of del
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering