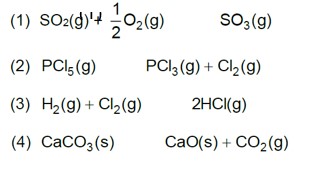
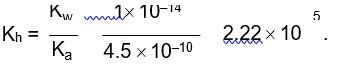7.20. One of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and CO2.
FeO (s) + CO (g) Fe (s) + CO2 (g); Kp = 0.265 atm at 1050K
What are the equilibrium partial pressures of CO and CO2 at 1050 K if the initial partial pressures are: pCO= 1.4 atm and pCO2= 0.80 atm?
7.20. One of the reactions that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and CO2.
FeO (s) + CO (g) Fe (s) + CO2 (g); Kp = 0.265 atm at 1050K
What are the equilibrium partial pressures of CO and CO2 at 1050 K if the initial partial pressures are: pCO= 1.4 atm and pCO2= 0.80 atm?
The initial partial pressures of CO and CO2 are 1.40 atm and 0.8. atm respectively.
The expression for the reaction quotient is:
Qp=PCO2 / PCO=0.80 / 1.4 = 0.571
As the value of the reaction quotient is greater than the value of the equilibrium constant, the reaction will move i
Similar Questions for you
0.01 M NaOH,
M = 1 * 10-2
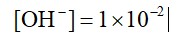
pOH = 2
pH = 2
Kp = Kc (RT)Dng
36 * 10–2 = Kc (0.0821 * 300)–1
Kc = 0.36 * 0.0821 * 300 = 8.86 » 9
A(g) ->B(g) + (g)
Initial moles n 0 &nbs
On increasing pressure, equilibrium moves in that direction where number of gaseous moles decreases.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering