8.2. What is the oxidation number of the underlined elements in each of the following and how do you rationalise your results?
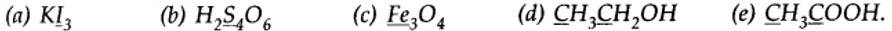
8.2. What is the oxidation number of the underlined elements in each of the following and how do you rationalise your results?
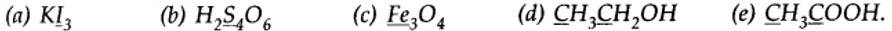
19 Views|Posted a year ago
Asked by Shiksha User
1 Answer
V
Answered by
a year ago
(a) In Kl3, since the oxidation number of K is +1, therefore, the average oxidation number of iodine = -1/3. But the oxidation number cannot be fractional. Therefore, we must consider its structure, K+ [I —I <— I]–. Here, a coordinate bond is formed between I2 molecule and I– ion. The oxidation number o
Similar Questions for you
Kindly go through the solution
(c) Li
Kindly go through the solution
(c) Al
Kindly go through the solution
(d) +6
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
7L
Reviews
|
1.9M
Answers
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering

