8.4. Fluorine reacts with ice and results in the change:
H2O(s) + F2 (g) ——> HF (g) + HOF (g)
Justify that this reaction is a redox reaction.
8.4. Fluorine reacts with ice and results in the change:
H2O(s) + F2 (g) ——> HF (g) + HOF (g)
Justify that this reaction is a redox reaction.
60 Views|Posted 12 months ago
Asked by Shiksha User
1 Answer
V
Answered by
12 months ago
Writing the O.N. of each atom above its symbol, we have,
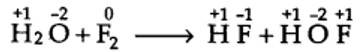
here, the O.N. of F decreases from 0 in F2 to -1 in HF and increases from 0 in F2 to +1 in HOF. Therefore, F2 is both reduced as well as oxidised. Thus, it is a redox reaction and more specifically, it is a disproportionation reaction
Similar Questions for you
Kindly go through the solution
(c) Li
Kindly go through the solution
(c) Al
Kindly go through the solution
(d) +6
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering

