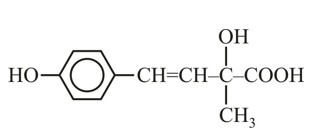
83 g of ethylene glycol dissolved in 625 g of water. The freezing point of the solution is________K. (Nearest integer).
[Use: Molal Freezing point depression constant of water = 1.86 K kg mol-1
Freezing point of water = 273K
Atomic masses: C : 12.0u, O : 16.0u, H : 1.0u]
83 g of ethylene glycol dissolved in 625 g of water. The freezing point of the solution is________K. (Nearest integer).
[Use: Molal Freezing point depression constant of water = 1.86 K kg mol-1
Freezing point of water = 273K
Atomic masses: C : 12.0u, O : 16.0u, H : 1.0u]
{for ethylene glycol I = 1, due to non-ionisable}
(the nearest integer)
Hence, freezing point of solution = 273 K – 4K = 269 K.
Similar Questions for you
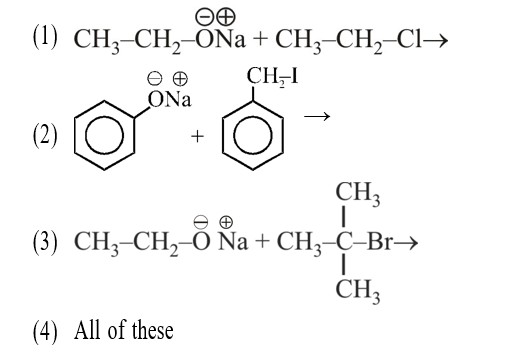
ΔG° = –RT * 2.303 log K
–nFE° = +RT * 2.303 log K
2 * 96500 * 0.295 = 8.314 * 298 * 2.303 log10 K
10 = log10 K = 1010
It has chiral centre and differently di substituted double bonded carbon atoms.
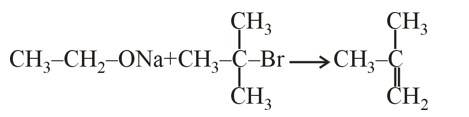
Rate of ESR ∝ No. of α – H (Hyperconjugation)
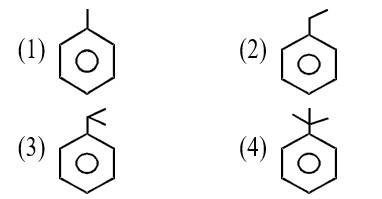
Cr3+ion is a most stable in aqueous solution due to. t2g half filled configuration
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Solutions 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering