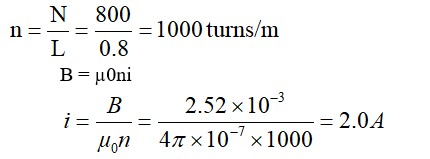A chloro compound “A”
(i) Forms aldehydes on ozonolysis followed by the hydrolysis.
(ii) When vaporized completely 1.53g of A, gives 448 mL of vapour at STP.
The number of carbon atoms in a molecule of compound A is________.
A chloro compound “A”
(i) Forms aldehydes on ozonolysis followed by the hydrolysis.
(ii) When vaporized completely 1.53g of A, gives 448 mL of vapour at STP.
The number of carbon atoms in a molecule of compound A is________.
Weight = 1.53 gm
V = 448 ml (STP)
Mole =
Molecular weight of compound A = 50 * 1.53 = 76.5 gram
Molecular weight 76.5 = CnH2n-1 Cl
or 12 n + 2n – 1 = 41
Molecular formula = C3H5Cl
Number of C- atoms = 3
Similar Questions for you
Photodiode in reverse bias mode is used as intensity measuring device.
Tertiary haloalkane does not undergo SN2 reaction
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering