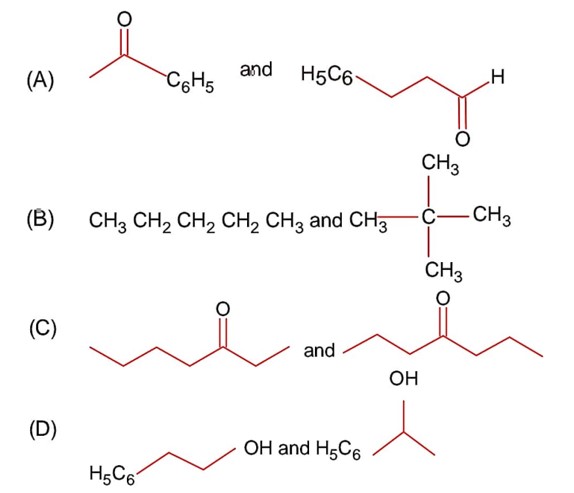
Among the following compounds, which one has the shortest bond?
Among the following compounds, which one has the shortest bond?
Option 1 - <p>Consider the following image</p>
<p><img ></p>
Option 2 - <p>Consider the following image</p>
<p><img ></p>
Option 3 - <p>Consider the following image</p>
<p><img ></p>
Option 4 - <p>Consider the following image</p>
<p><img ></p>
2 Views|Posted 10 months ago
Asked by Shiksha User
1 Answer
A
Answered by
10 months ago
Correct Option - 3
Detailed Solution:
Sol. and both have 4 unpaired electrons.
Similar Questions for you
4-ethoxycarbonylpent-3-enoic acid
In metamers, distribution of alkyl groups are changed with respect to polyvalent functional groups.
Sodium fusion extract is boiled with conc. HNO3 to remove NaCN and Na2S.
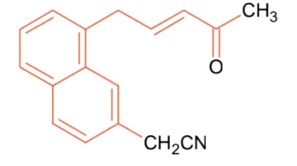
Number of sp2 hybridized carbon in A = 2
Kindly consider the following figure
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry NCERT Exemplar Solutions Class 12th Chapter Twelve 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering