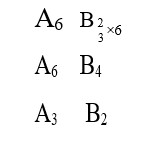An LPG cylinder contains gas at a pressure of 300 kPa at 27°C. The cylinder can withstand the pressure of 1.2*10? Pa. The room in which the cylinder is kept catches fire. The minimum temperature at which the bursting of cylinder will take place is __________ °C. (Nearest integer)
An LPG cylinder contains gas at a pressure of 300 kPa at 27°C. The cylinder can withstand the pressure of 1.2*10? Pa. The room in which the cylinder is kept catches fire. The minimum temperature at which the bursting of cylinder will take place is __________ °C. (Nearest integer)
P? = 300 kPa = 3 * 10? Pa = 3 bar
T? = 300 K
P? = 1.2 * 10? Pa = 12 * 10? Pa = 12 bar
T? =?
P? /T? = P? /T?
3/300 = 12/T?
T? = (12 * 300)/3 K = 1200 K
∴ T in °C = 1200 - 273 = 927°C
Similar Questions for you
CH3COOH + NaOH → CH3COONa + H2O
ΔH = –50.6 kJ/mol
NaOH + SA [HCl] → NaCl + H2O
ΔH = –55.9 kJ/mol
the value of ΔH for ionisation of CH3COOH
⇒ ΔH = +55.9 – 50.6
5.3 kJ/mol
Kindly consider the solution
Fact.
Kindly go through the solution
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering