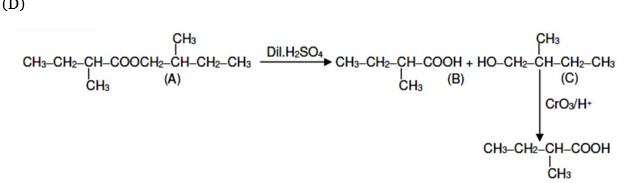
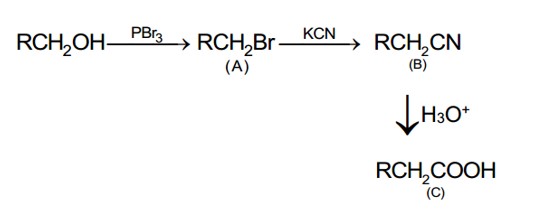
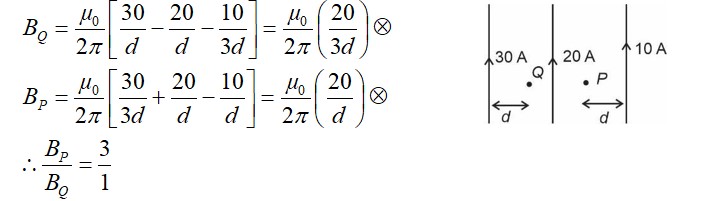An organic compound [A], molecular formula C??H??O?, was hydrolyzed with dilute sulphuric acid to give a carboxylic acid [B] and an alcohol [C]. Oxidation of [C] with CrO? – H?SO? produced [B]. Which of the following structures are not possible for [A]?
An organic compound [A], molecular formula C??H??O?, was hydrolyzed with dilute sulphuric acid to give a carboxylic acid [B] and an alcohol [C]. Oxidation of [C] with CrO? – H?SO? produced [B]. Which of the following structures are not possible for [A]?
(B) contains eight carbon atoms whereas the molecular formula C? H? O? contains ten carbon atoms.
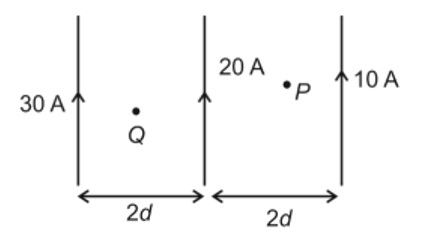
Similar Questions for you
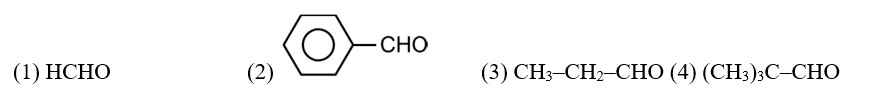
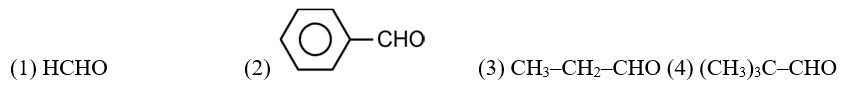
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering