An organic compound (A) (Molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) And an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) On dehydration gives but-1-ene. Write equations for the reactions involved.(Intermediate)
An organic compound (A) (Molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) And an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) On dehydration gives but-1-ene. Write equations for the reactions involved.(Intermediate)
ANSWER: Given compound is having 8 carbons, and on reaction with chromic acid C is converted back into B, as chromic acid reaction couldn't add any carbon hence both alcohol and acid must contain 4 carbons and it is given in the question that on dehydration C will give but-1-ene so alcohol will be b
Similar Questions for you
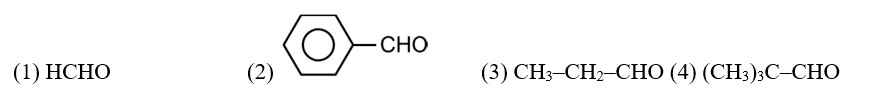
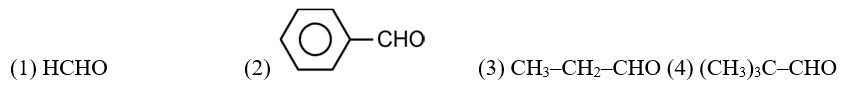
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Aldehydes, Ketones and Carboxylic Acids 2021
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering