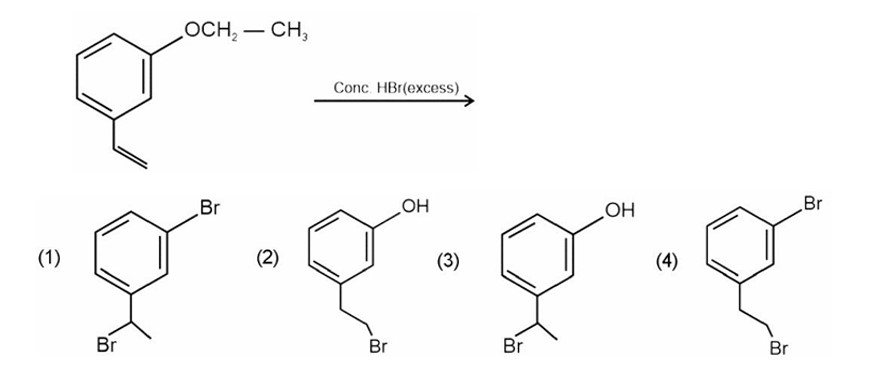
Arrange the halogens F2, Cl2, Br2, I2, in order of their increasing reactivity with alkanes.
(i) I2< Br2< Cl2< F2
(ii) Br2< Cl2< F2< I2
(iii) F2< Cl2< Br2< I2
(iv) Br2< I2 < Cl2< F2
Arrange the halogens F2, Cl2, Br2, I2, in order of their increasing reactivity with alkanes.
(i) I2< Br2< Cl2< F2
(ii) Br2< Cl2< F2< I2
(iii) F2< Cl2< Br2< I2
(iv) Br2< I2 < Cl2< F2
This is a multiple choice answer as classified in NCERT Exemplar
(i) I2< Br2< Cl2< F2
The reactivity of halogens with alkanes depends on the electronegativity i.e. higher the electronegativity higher would be the reaction. As down the group reactivity decrease so thus its reactivity.
Similar Questions for you
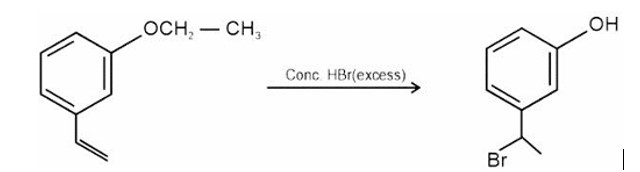
HBr adds to alkene in accordance with Markovnikov's rule.
Delocalisation of
To study Hydrocarbons for NEET, you can use the Hydrocarbons Class 11th NCERT solutions PDF.
Alkanes, Alkenes, Alkynes, and Aromatics hydrocarbons are the four main hydrocarbons.
Hydrocarbons are organic compounds made of only carbon and hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Thirteen 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering