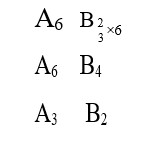For the reaction . At a given time, the composition of reaction mixture is: . Then, which of the following is correct?
For the reaction . At a given time, the composition of reaction mixture is: . Then, which of the following is correct?
Option 1 - <p><span lang="EN-IN">Reaction is at equilibrium.</span></p>
Option 2 - <p><span lang="EN-IN">Reaction has a tendency to go in forward direction.</span></p>
Option 3 - <p><span lang="EN-IN">Reaction has a tendency to go in backward direction.</span></p>
Option 4 - <p><span lang="EN-IN">Reaction has gone to completion in forward direction.</span></p>
6 Views|Posted 8 months ago
Asked by Shiksha User
1 Answer
P
Answered by
8 months ago
Correct Option - 3
Detailed Solution:
At a given time is to be calculated and been compared with .
As , so reaction has a tendency to move backward.
Similar Questions for you
CH3COOH + NaOH → CH3COONa + H2O
ΔH = –50.6 kJ/mol
NaOH + SA [HCl] → NaCl + H2O
ΔH = –55.9 kJ/mol
the value of ΔH for ionisation of CH3COOH
⇒ ΔH = +55.9 – 50.6
5.3 kJ/mol
Kindly consider the solution
Fact.
Kindly go through the solution
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Classification of Elements and Periodicity in Properties 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering