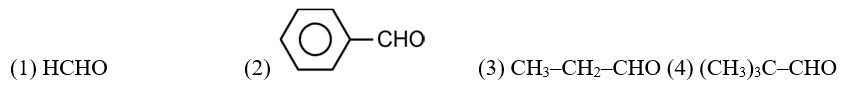
Give plausible explanation for each of the following:
(i) Cyclohexanone forms cyanohydrin in good yield but 2,2,6-trimethylcyclohexanone does not.
(ii) There are two –NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
(iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Give plausible explanation for each of the following:
(i) Cyclohexanone forms cyanohydrin in good yield but 2,2,6-trimethylcyclohexanone does not.
(ii) There are two –NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
(iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
1. Cyclohexanone forms cyanohydrin in good yield because the ketonic group has very less steric hindrance at both the ortho position but 2,2,6 tri methyl cyclohexanone have high steric hindrance which reduces the attack from CN nucleophile.
Similar Questions for you
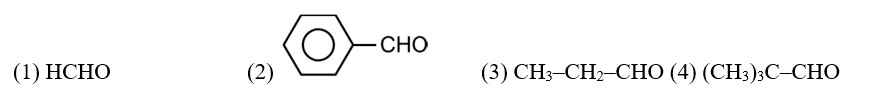
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Aldehydes, Ketones and Carboxylic Acids 2021
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering