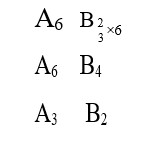Is knowing just the atomic number or just the mass number enough to identify isotopes and isobars?
Is knowing just the atomic number or just the mass number enough to identify isotopes and isobars?
1 Views|Posted 8 months ago
Asked by Shiksha User
1 Answer
S
Answered by
8 months ago
Correctly identifying isotopes and isobars requires knowing both the atomic and mass numbers. Relying on only one is a common error.
- Isotopes: Same element (atomic number), different mass.
- Isobars: Different elements (atomic number), same mass.
Similar Questions for you
CH3COOH + NaOH → CH3COONa + H2O
ΔH = –50.6 kJ/mol
NaOH + SA [HCl] → NaCl + H2O
ΔH = –55.9 kJ/mol
the value of ΔH for ionisation of CH3COOH
⇒ ΔH = +55.9 – 50.6
5.3 kJ/mol
Kindly consider the solution
Fact.
Kindly go through the solution
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering