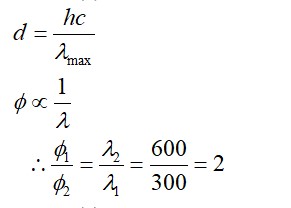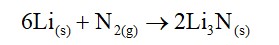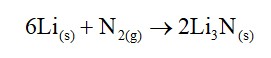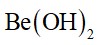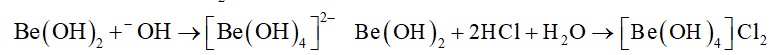Lithium hydride can be used to prepare other useful hydrides. Beryllium hydride is one of them. Suggest a route for the preparation of beryllium hydride starting from lithium hydride. Write chemical equations involved in the process.
Lithium hydride can be used to prepare other useful hydrides. Beryllium hydride is one of them. Suggest a route for the preparation of beryllium hydride starting from lithium hydride. Write chemical equations involved in the process.
This is a long answer type question as classified in NCERT Exemplar
Beryllium being least reactive does not form hydride by direct heating with dihydrogen. It is prepared by reacting it with lithium aluminum hydride. The reactions are shown below.
8LiH + Al2Cl6→2LiAlH4+6LiCl
2BeCl2+LiAlH4→2BeH2+LiCl+Al
Similar Questions for you
Li+ has the highest hydration enthalpy.
Hence it is most hydrated
Therefore, Correct order of hydrated radii is Cs+ < Rb+ < K+ < Na+ < Li+
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Ten 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering